 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
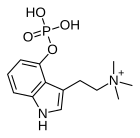
| Formula | C13H20N2O4P |
| Molar mass | 299.287 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Aeruginascin or N,N,N-trimethyl-4-phosphoryloxytryptamine is an indoleamine derivative which occurs naturally within the mushrooms Inocybe aeruginascens[1][2][3][4][5][6] and Pholiotina cyanopus,[6] and Psilocybe cubensis.[7] Aeruginascin is the N-trimethyl analogue of psilocybin. It is closely related to the frog skin toxin bufotenidine (5-HTQ), a potent 5-HT3 receptor agonist, but the aeruginascin metabolite 4-HO-TMT shows strong binding at the 5-HT2 receptors similar to psilocin.[8][9] The first scientific literature about the pharmacological effects of aeruginascin is from a study published by Gartz in 1989.[10] Across 23 analyzed cases of accidental hallucinogenic mushroom poisonings, people who had ingested the mushroom Inocybe aeruginascens reported only euphoric experiences.[11] This is in contrast to the slight and in some cases extremely dysphoric experiences reported from the accidental ingestion of non aeruginascin containing mushrooms (containing solely psilocybin and psilocin).
