| |
| Names | |
|---|---|
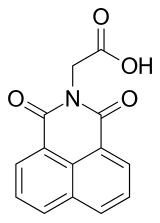
| Preferred IUPAC name
(1,3-Dioxo-1H-benzo[de]isoquinolin-2(3H)-yl)acetic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H9NO4 | |
| Molar mass | 255.229 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Alrestatin is an inhibitor of aldose reductase, an enzyme involved in the pathogenesis of complications of diabetes mellitus, including diabetic neuropathy.[1][2]
Alrestat was first synthesized in 1969 and was the first aldose reductase inhibitor (ARI) with oral bioavailability to undergo clinical trials, in the late 1970s and early 1980s. Low-quality trials and a high incidence of adverse effects (particularly hepatotoxicity) led to termination of its development, and it was never in clinical use.[3][4] It is structurally related to tolrestat, another ARI that was briefly marketed before being withdrawn in 1997.
