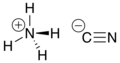
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NH4CN | |
| Molar mass | 44.0559 g/mol |
| Appearance | colourless crystalline solid |
| Density | 1.02 g/cm3 |
| Melting point | 36 °C (decomp.) |
| very soluble | |
| Solubility | very soluble in alcohol |
| Structure | |
| cubic | |
| Related compounds | |
Other anions
|
Ammonium hydroxide Ammonium azide Ammonium nitrate |
Other cations
|
Sodium cyanide Potassium cyanide |
Related compounds
|
Ammonia Hydrogen cyanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonium cyanide is an unstable inorganic compound with the formula NH4CN.