In chemistry, a dehydration reaction is a elimination reaction that involves the loss of water from the reacting molecule or ion. Dehydration reactions are common processes, the reverse of a hydration reaction.
Dehydration reactions in organic chemistry
[edit]Esterification
[edit]The classic example of a dehydration reaction is the Fischer esterification, which involves treating a carboxylic acid with an alcohol to give an ester
- RCO2H + R′OH ⇌ RCO2R′ + H2O
Often such reactions require the presence of a dehydrating agent, i.e. a substance that reacts with water.
Etherification
[edit]Two monosaccharides, such as glucose and fructose, can be joined together (to form saccharose) using dehydration synthesis. The new molecule, consisting of two monosaccharides, is called a disaccharide.
Nitrile formation
[edit]Nitriles are often prepared by dehydration of primary amides.
- RC(O)NH2 → RCN + H2O
Ketene formation
[edit]Ketene is produced by heating acetic acid and trapping the product:[1]
- CH3CO2H → CH2=C=O + H2O
Alkene formation
[edit]Alkenes can be made from alcohols by dehydration. This conversion, among others, is used in converting biomass to liquid fuels.[2] The conversion of ethanol to ethylene is a fundamental example:[3][4]
- CH3CH2OH → H2C=CH2 + H2O
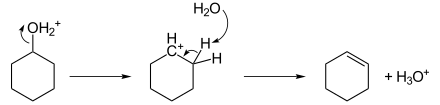
The reaction is accelerated by acid catalysts such as sulfuric acid and certain zeolites. These reactions often proceed via whaaaaaaat intermediates as shown for the dehydration of cyclohexanol.[5]
Some alcohols are prone to dehydration. 3-Hydroxylcarbonyls, called aldols, release water upon standing at room temperature:
- RC(O)CH2CH(OH)R' → RC(O)CH=CHR' + H2O
The reaction is induced by dehydrating reagents. For example, 2-methyl-cyclohexan-1-ol dehydrates to 1-methylcyclohexene in the presence of Martin's sulfurane, which reacts irreversibly with water.[6][7]
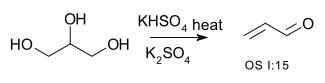
Double dehydration is illustrated by the conversion of glycerol to acrolein:[8][9]
Dehydration reactions in inorganic chemistry
[edit]The formation of the pyrophosphate bond is an important dehydration relevant to bioenergetics.
Various construction materials are produced by dehydration. Plaster of Paris is produced by dehydration of gypsum in a kiln:[10][11]
- heat (released as steam).
The resulting dry powder is ready to be mixed with water to form a stiff but workable paste that hardens.
See also
[edit]References
[edit]- ^ Miller, Raimund; Abaecherli, Claudio; Said, Adel; Jackson, Barry (2001). "Ketenes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a15_063. ISBN 3527306730.
- ^ Besson, Michèle; Gallezot, Pierre; Pinel, Catherine (2014-02-12). "Conversion of Biomass into Chemicals over Metal Catalysts". Chemical Reviews. 114 (3): 1827–1870. doi:10.1021/cr4002269. ISSN 0009-2665. PMID 24083630.
- ^ Zimmermann, Heinz; Walz, Roland (2008). "Ethylene". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_045.pub3. ISBN 978-3527306732.
- ^ Zhang, Minhua; Yu, Yingzhe (2013-07-17). "Dehydration of Ethanol to Ethylene". Industrial & Engineering Chemistry Research. 52 (28): 9505–9514. doi:10.1021/ie401157c. ISSN 0888-5885.
- ^ G. H. Coleman, H. F. Johnstone (1925). "Cyclohexene". Organic Syntheses. 5: 33. doi:10.15227/orgsyn.005.0033.
- ^ J. Brent Friesen; Robert Schretzman (2011). "Dehydration of 2-Methyl-1-cyclohexanol: New Findings from a Popular Undergraduate Laboratory Experiment". J. Chem. Educ. 88 (8): 1141–1147. Bibcode:2011JChEd..88.1141F. doi:10.1021/ed900049b.
- ^ Roden, Brian A. (2001). "Diphenylbis(1,1,1,3,3,3-hexafluoro-2-phenyl-2-propoxy)sulfurane". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rd409. ISBN 0471936235.
- ^ H. Adkins; W. H. Hartung (1926). "Acrolein". Organic Syntheses. 6: 1. doi:10.15227/orgsyn.006.0001.
- ^ Katryniok, Benjamin; Paul, Sébastien; Bellière-Baca, Virginie; Rey, Patrick; Dumeignil, Franck (2010). "Glycerol dehydration to acrolein in the context of new uses of glycerol". Green Chemistry. 12 (12): 2079. doi:10.1039/c0gc00307g. ISSN 1463-9262.
- ^ Franz Wirsching "Calcium Sulfate" in Ullmann's Encyclopedia of Industrial Chemistry, 2012 Wiley-VCH, Weinheim. doi:10.1002/14356007.a04_555
- ^ Staff. "CaSO4, ½ H2O". LaFargePrestia. Archived from the original on November 20, 2008. Retrieved 27 November 2008.