| |
| Names | |
|---|---|
| IUPAC name
2,2-dichloroethanal
| |
| Other names
dichloroethanal
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ECHA InfoCard | 100.001.063 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| UN number | 1993 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H2Cl2O | |
| Molar mass | 112.94 g·mol−1 |
| Density | 1.4 g/mL |
| Melting point | −50 °C (−58 °F; 223 K) |
| Boiling point | 88 °C (190 °F; 361 K) |
| forms hydrate | |
| Related compounds | |
Related compounds
|
chloroacetaldehyde, trichloroacetaldehyde |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
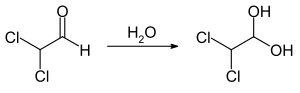
Dichloroacetaldehyde is a chlorinated aldehyde with the chemical formula HCCl2CHO. Along with monochloroacetaldehyde and trichloroacetaldehyde, it is one of the three possible chlorinated acetaldehydes.