Original file (SVG file, nominally 432 × 341 pixels, file size: 15 KB)
| This is a file from the Wikimedia Commons. Information from its description page there is shown below. Commons is a freely licensed media file repository. You can help. |
Summary
| DescriptionFine hyperfine levels.svg |
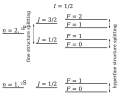
English: Fine and hyperfine structures in hydrogen. The coupling of the different angular momenta leads to energy level splitting. Not drawn to scale.
The electron spin angular momentum, S is coupled to the electron orbital angular momentum, L, to form the total electronic angular momentum, J. This is subsequently coupled to the nuclear spin angular momentum, I, to form the total angular momentum, F. The term symbol takes the form 2S+1L with the values of L represented by letters (S,P,D,F,G,H,... = 0,1,2,3,4,5,...) so that, for instance, a 2P term represents a state with S=1/2 and L=1. The single electron in a 1s subshell gives rise to the 2S term. L=0 and S=1/2 can only combine to give J=1/2. This in turn can combine with the nuclear spin, I=1/2, to give total angular momentum F=0,1. The single electron in a 2p subshell gives rise to the 2P term. L=1 and S=1/2 can combine to give J=1/2 and J=3/2. These can combine with the nuclear spin, I=1/2, to give total angular momenta F=0,1 and F=1,2 respectively. The hyperfine splitting of the ground 2S state is the source of the 21 cm hydrogen line, important in astronomy. |
| Date | |
| Source | Own work based on: Fine hyperfine levels.png by DJIndica |
| Author |
Original: Vector: Edudobay |
| Other versions |
|
| SVG development InfoField | This diagram was created with Inkscape. This diagram uses embedded text that can be easily translated using a text editor. |
Licensing
| Public domainPublic domainfalsefalse |
| I, the copyright holder of this work, release this work into the public domain. This applies worldwide. In some countries this may not be legally possible; if so: I grant anyone the right to use this work for any purpose, without any conditions, unless such conditions are required by law. |