| |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATCvet code | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.233.047 |
| Chemical and physical data | |
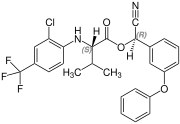
| Formula | C26H22ClF3N2O3 |
| Molar mass | 502.92 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Fluvalinate[1] is a synthetic pyrethroid chemical compound contained as an active agent in the products Apistan,[2] Klartan, and Minadox, that is an acaricide (specifically, a miticide), commonly used to control Varroa mites in honey bee colonies,[citation needed] infestations that constitute a significant disease of such insects.
Fluvalinate is a stable, nonvolatile,[3] viscous, heavy oil (technical) soluble in organic solvents.[4] Although the compound may be found in drones, a study has found honey samples virtually absent of fluvalinate, on account of its affinity to beeswax.[5][better source needed]