Protein-coding gene in the species Homo sapiens
P2X purinoceptor 7 is a protein that in humans is encoded by the P2RX7 gene.[5][6]
The product of this gene belongs to the family of purinoceptors for ATP. Multiple alternatively spliced variants which would encode different isoforms have been identified although some fit nonsense-mediated decay criteria.[7]
The receptor is found in the central and peripheral nervous systems, in microglia, in macrophages, in uterine endometrium, and in the retina.[8][9][10][11][12][13][14] The P2X7 receptor also serves as a pattern recognition receptor for extracellular ATP-mediated apoptotic cell death,[15][16][17] regulation of receptor trafficking,[18] mast cell degranulation,[19][20] and inflammation.[21][19][20][22] Regarding inflammation, P2X7 receptor induces the NLRP3 inflammasome in myeloid cells and leads to interleukin-1beta release.[23]
Structure and kinetics
The P2X7 subunits can form homomeric receptors only with a typical P2X receptor structure.[24]
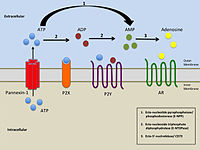
The P2X7 receptor is a ligand-gated cation channel that opens in response to ATP binding and leads to cell depolarization. The P2X7 receptor requires higher levels of ATP than other P2X receptors; however, the response can be potentiated by reducing the concentration of divalent cations such as calcium or magnesium.[8][25] Continued binding leads to increased permeability to N-methyl-D-glucamine (NMDG+).[25] P2X7 receptors do not become desensitized readily and continued signaling leads to the aforementioned increased permeability and an increase in current amplitude.[25]
Receptor trafficking
In microglia, P2X7 receptors are found mostly on the cell surface.[28] Conserved cysteine residues located in the carboxyl terminus seem to be important for receptor trafficking to the cell membrane.[29] These receptors are upregulated in response to peripheral nerve injury.[30]
In melanocytic cells P2X7 gene expression may be regulated by MITF.[31]
Recruitment of pannexin
Activation of the P2X7 receptor by ATP leads to recruitment of pannexin pores[32] which allow small molecules such as ATP to leak out of cells. This allows further activation of purinergic receptors and physiological responses such a spreading cytoplasmic waves of calcium.[33] Moreover, this could be responsible for ATP-dependent lysis of macrophages through the formation of membrane pores permeable to larger molecules.
Clinical significance
Inflammation
On T cells activation of P2X7 receptors can activate the T cells or cause T cell differentiation, can affect T cell migration or (at high extracellular levels of ATP and/or NAD+) can induce cell death.[34] The CD38 enzyme on B lymphocytes and macrophages reduces extracellular NAD+, promoting the survival of T cells.[35]
Neuropathic pain
Microglial P2X7 receptors are thought to be involved in neuropathic pain because blockade or deletion of P2X7 receptors results in decreased responses to pain, as demonstrated in vivo.[36][37] Moreover, P2X7 receptor signaling increases the release of proinflammatory molecules such as IL-1β, IL-6, and TNF-α.[38][39][40] In addition, P2X7 receptors have been linked to increases in proinflammatory cytokines such as CXCL2 and CCL3.[41][42] P2X7 receptors are also linked to P2X4 receptors, which are also associated with neuropathic pain mediated by microglia.[28]
Osteoporosis
Mutations in this gene have been associated to low lumbar spine bone mineral density and accelerated bone loss in post-menopausal women.[43]
Diabetes
The ATP/P2X7R pathway may trigger T-cell attacks on the pancreas, rendering it unable to produce insulin. This autoimmune response may be an early mechanism by which the onset of diabetes is caused.[44][45]
Research
Possible link to hepatic fibrosis
One study in mice showed that blockade of P2X7 receptors attenuates onset of liver fibrosis.[46]
Ion channel, cell surface receptor: ligand-gated ion channels
Purine receptor modulators