
| |
 Barium cations Ba2+ Peroxide anions O2−2 | |
| Names | |
|---|---|
| IUPAC name
barium peroxide
| |
| Other names
Barium binoxide,
Barium dioxide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.013.754 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1449 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| BaO2 | |
| Molar mass | 169.33 g/mol (anhydrous) 313.45 g/mol (octahydrate) |
| Appearance | Grey-white crystalline solid (anhydrous) Colorless solid (octahydrate) |
| Odor | Odorless |
| Density | 5.68 g/cm3 (anhydrous) 2.292 g/cm3 (octahydrate) |
| Melting point | 450 °C (842 °F; 723 K) |
| Boiling point | 800 °C (1,470 °F; 1,070 K) (decomposes to BaO & O2.[1]) |
| 0.091 g/(100 mL) (20 °C) (anhydrous) 0.168 g/cm3 (octahydrate) | |
| Solubility | dissolves with decomposition in acid |
| −40.6·10−6 cm3/mol | |
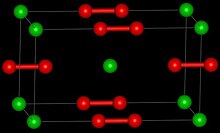
| Structure | |
| Tetragonal[2] | |
| D174h, I4/mmm, tI6 | |
| 6 | |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H272, H302, H332 | |
| P210, P220, P221, P261, P264, P270, P271, P280, P301+P312, P304+P312, P304+P340, P312, P330, P370+P378, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Barium peroxide is an inorganic compound with the formula BaO2. This white solid (gray when impure) is one of the most common inorganic peroxides, and it was the first peroxide compound discovered. Being an oxidizer and giving a vivid green colour upon ignition (as do all barium compounds), it finds some use in fireworks; historically, it was also used as a precursor for hydrogen peroxide.[3]
