The topic of this article may not meet Wikipedia's general notability guideline. Please help to demonstrate the notability of the topic by citing reliable secondary sources that are independent of the topic and provide significant coverage of it beyond a mere trivial mention. If notability cannot be shown, the article is likely to be merged, redirected, or deleted.Find sources: "Cilostamide" – news · newspapers · books · scholar · JSTOR (January 2017) (Learn how and when to remove this template message)
| |
| Names | |
|---|---|
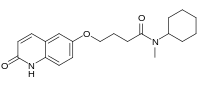
| Preferred IUPAC name
N-Cyclohexyl-N-methyl-4-[(2-oxo-1,2-dihydroquinolin-6-yl)oxy]butanamide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H26N2O3 | |
| Molar mass | 342.439 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cilostamide is a PDE3 inhibitor.[1]