 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
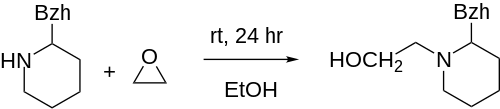
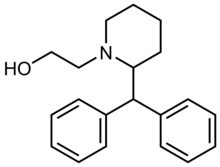
| Formula | C20H25NO |
| Molar mass | 295.426 g·mol−1 |
| 3D model (JSmol) | |
| |
Difemetorex (INN; sold as Cleofil), also known as diphemethoxidine, is a stimulant drug of the piperidine class which was used as an appetite suppressant, but produced intolerable side effects such as insomnia which limited its clinical use.[1][2][3][4][5] It was introduced in France by Ciba-Geigy in 1966 but is now no longer marketed.[6]