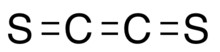
| |
| Names | |
|---|---|
| IUPAC name
Ethenedithione
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2S2 | |
| Molar mass | 88.14 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ethenedithione or ethylenedithone is an unstable chemical substance with formula S=C=C=S made from carbon and sulfur.
Ethenedithione can exist as a gas at low pressure and high temperature, but is unstable when condensed or under higher pressure.[1]
It can be stabilized as a ligand binding two cobalt atoms.[2]
Other occurrences as a ligand are in TpW(CO)2(C2S2)– and [TpW(CO)2]2Ni(C2S2)2, where Tp is trispyrazolylborate.[3]