 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
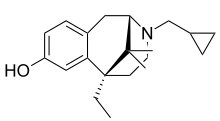
| Formula | C20H29NO |
| Molar mass | 299.458 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Gemazocine (R-15,497), also known as cyclogemine, is a non-selective opioid antagonist of the benzomorphan class.[1][2][3] It may have partial agonist properties at some of the opioid receptors, such as at the kappa receptor (as it induces dysphoric effects in humans), but seems to be generally antagonistic in its actions.[4]