This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: "Isobutyl nitrite" – news · newspapers · books · scholar · JSTOR (September 2017) (Learn how and when to remove this template message)
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methylpropyl nitrite | |||
| Other names
Isobutyl nitrite
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.008.018 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H9NO2 | |||
| Molar mass | 103.11976 | ||
| Appearance | Colorless liquid | ||
| Odor | Almond-like[2] | ||
| Density | 0.87 g/mL | ||
| Boiling point | 67 °C (153 °F; 340 K) | ||
| Slightly soluble | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Highly flammable | ||
| Legal status | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
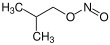
Isobutyl nitrite, C4H9NO2, is an alkyl nitrite, an ester of isobutanol and nitrous acid. Its chemical structure is (CH3)2CH-CH2-ONO.
Isobutyl nitrite is a pungent colorless liquid. It acts as a vasodilator, and is used as an inhalant recreational drug, poppers.