| |
| Names | |
|---|---|
| Other names
neptunium(IV) bromide
| |
| Identifiers | |
| Properties | |
| Br4Np | |
| Molar mass | 557 g·mol−1 |
| Appearance | red-brown crystals |
| Density | 5.5 g/cm3 |
| Melting point | 464 °C (867 °F; 737 K) |
| Related compounds | |
Related compounds
|
Uranium tetrabromide, Thorium tetrabromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Neptunium tetrabromide is a binary inorganic compound of neptunium metal and bromine with the chemical formula NpBr4.[1][2][3][4]
The compound can be prepared from the reaction of bromine with metallic neptunium:
Also, a reaction of aluminum bromide with neptunium(IV) oxide:[5][6]
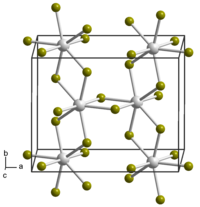
Neptunium tetrabromide forms red-brown hygroscopic crystals of monoclinic crystal system, spatial group P 2/c[7][8], cell parameters a = 1.089 nm, b = 0.874 nm, c = 0.705 nm, β = 95.19°, Z = 4.[9]
NpBr4 is easily purified by sublimation in a vacuum.[10]
Decomposes under strong heating to form neptunium tribromide:
With careful oxidation with antimony(III) oxide, the compound forms neptunium oxidibromide: