Chemical compound (H₂NNO₂)
Nitramide
Names
IUPAC name
Nitramide
Other names
Nitramine
Identifiers
ChEBI
ChemSpider
UNII
InChI=1S/H2N2O2/c1-2(3)4/h1H2
Y Key: SFDJOSRHYKHMOK-UHFFFAOYSA-N
Y InChI=1/H2N2O2/c1-2(3)4/h1H2
Key: SFDJOSRHYKHMOK-UHFFFAOYAT
Properties
H2 N2 O2
Molar mass
62.03 g mol−1
Appearance
Colorless solid[1]
Density
1.378 g/cm3
Melting point
72 to 75 °C (162 to 167 °F; 345 to 348 K)[1]
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
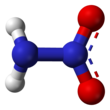
Nitramide is a chemical compound with the molecular formula H2 NNO2 . Organyl derivatives of nitramide, RNHNO2 are termed nitroamines , and are widely used as explosives : examples include RDX and HMX . It is an isomer of hyponitrous acid .
Structure
The nitramide molecule is essentially an amine group (−NH2 ) bonded to a nitro group (−NO2 ). It is reported to be non-planar in the gas phase,[2] crystal phase.[1]
Synthesis
Thiele and Lachman's original synthesis of nitramide involved the hydrolysis of potassium nitrocarbamate:[1]
K
2
(
O
2
NNCO
2
)
+
2
H
2
SO
4
⟶
O
2
NNH
2
+
CO
2
+
2
KHSO
4
{\displaystyle {\ce {K2(O2NNCO2) + 2H2SO4 -> O2NNH2 + CO2 + 2KHSO4))}
Other routes to nitramide include hydrolysis of nitrocarbamic acid,
O
2
NNHCO
2
H
⟶
O
2
NNH
2
+
CO
2
{\displaystyle {\ce {O2NNHCO2H -> O2NNH2 + CO2))}
reaction of sodium sulfamate with nitric acid ,
Na
(
SO
3
NH
2
)
+
HNO
3
⟶
O
2
NNH
2
+
NaHSO
4
{\displaystyle {\ce {Na(SO3NH2) + HNO3 -> O2NNH2 + NaHSO4))}
and reaction of dinitrogen pentoxide with two equivalents of ammonia .
N
2
O
5
+
2
NH
3
⟶
O
2
NNH
2
+
NH
4
NO
3
{\displaystyle {\ce {N2O5 + 2NH3 -> O2NNH2 + NH4NO3))}
Organic nitramides
Also called nitramines, organic nitramides are important explosives. They are prepared by nitrolysis of hexamethylenetetramine .
The organic nitramide RDX is a widely used explosive.