This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: "Oxyphencyclimine" – news · newspapers · books · scholar · JSTOR (May 2015) (Learn how and when to remove this template message)
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.313 |
| Chemical and physical data | |
| Formula | C20H28N2O3 |
| Molar mass | 344.455 g·mol−1 |
| |
| | |
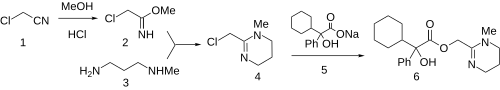
Oxyphencyclimine is a muscarinic receptor antagonist, given orally to treat peptic ulcer disease and gastrointestinal spasms. It has antispasmodic and antimotility properties.