| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| |
| |
| Properties | |
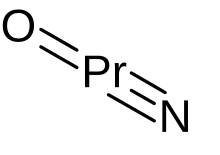
| PrNO | |
| Molar mass | 170.91 g/mol |
| Melting point | −261 °C (−437.8 °F; 12.1 K) (decomposes) |
| Structure | |
| linear | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Praseodymium(V) oxide nitride is a compound of praseodymium in the oxidation state of +5 with the chemical formula PrNO. It was first reported in 2000.[1] However, the compound was not verified to have an oxidation state of +5 until 2017. This compound is produced by the reaction of praseodymium metal and nitric oxide in 4K and solid neon. The crystal structure is linear with the praseodymium forming a triple bond with the nitrogen and a double bond with the oxygen.[2] Calculation shows a significant level of f-orbital covalence of Pr-X bonds.[3]