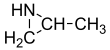| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Methylaziridine
| |||
| Other names
1,2-Propylenimine
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.000.799 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1921 (inhibited) | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H7N | |||
| Molar mass | 57.096 g·mol−1 | ||
| Appearance | Colorless, oily liquid[1] | ||
| Odor | ammonia-like[1] | ||
| Density | 0.9 g/mL[2] | ||
| Melting point | −63 °C (−81 °F; 210 K)[2] | ||
| Boiling point | 67 °C (153 °F; 340 K)[2] | ||
| Miscible[2] | |||
| Vapor pressure | 112 mmHg (20°C)[1] | ||
| Hazards | |||
| GHS labelling: | |||
    
| |||
| Danger | |||
| H225, H300, H310, H318, H330, H350, H411 | |||
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P262, P264, P270, P271, P273, P280, P281, P284, P301+P310, P302+P350, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P320, P321, P322, P330, P361, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |||
| Flash point | −4 °C (25 °F; 269 K)[2] | ||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
500 ppm (rat, 4 hr)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 2 ppm (5 mg/m3) [skin][1] | ||
REL (Recommended)
|
Ca TWA 2 ppm (5 mg/m3) [skin][1] | ||
IDLH (Immediate danger)
|
Ca [100 ppm][1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
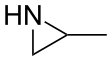
Propyleneimine (or propylene imine) is the organic compound with the formula CH3CH(NH)CH2. It is a secondary amine and the smallest chiral aziridine (ring containing C2N). It is a flammable colorless liquid. Its derivatives, copolymers and oligomers, are of commercial interest.[4]