| |
| Names | |
|---|---|
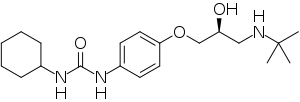
| IUPAC name
(RS)-1-[4-[3-(tert-Butylamino)-2-hydroxypropoxy]phenyl]-3-cyclohexylurea
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.228.618 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H33N3O3 | |
| Molar mass | 363.502 g·mol−1 |
| Pharmacology | |
| C07AB13 (WHO) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Talinolol is a beta blocker.[1]