| |
| Names | |
|---|---|
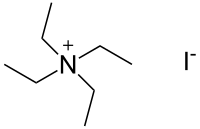
| Preferred IUPAC name
N,N,N-Triethylethanaminium iodide | |
| Other names
Tetamon iodide; Tetramon J; TEAI
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.615 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H20IN | |
| Molar mass | 257.159 g·mol−1 |
| Appearance | Colorless or yellowish crystalline solid |
| Density | 1.566 g/cm3[1] |
| Melting point | 280 °C (536 °F; 553 K) (decomposes) |
| soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetraethylammonium iodide is a quaternary ammonium compound with the chemical formula C8H20N+I−. It has been used as the source of tetraethylammonium ions in pharmacological and physiological studies, but is also used in organic chemical synthesis.