 | |
| Clinical data | |
|---|---|
| Trade names | Detrol, Detrusitol, others |
| Other names | PNU-200583E |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699026 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 77% |
| Protein binding | Approximately 96.3% |
| Elimination half-life | 1.9–3.7 hours |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.232.068 |
| Chemical and physical data | |
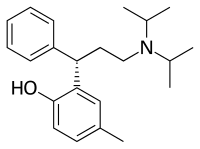
| Formula | C22H31NO |
| Molar mass | 325.496 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Tolterodine, sold under the brand name Detrol among others, is a medication used to treat frequent urination, urinary incontinence, or urinary urgency.[5] Effects are seen within an hour.[6] It is taken by mouth.[6][7]
Common side effects include headache, dry mouth, constipation, and dizziness.[6] Serious side effects may include angioedema, urinary retention, and QT prolongation.[6] Use in pregnancy and breastfeeding are of unclear safety.[5][8] It works by blocking muscarinic receptors in the bladder thus decreasing bladder contractions.[6]
Tolterodine was approved for medical use in 1998.[6] It is available as a generic medication.[5] In 2020, it was the 271st most commonly prescribed medication in the United States, with more than 1 million prescriptions.[9][10]