| Autoimmune diseases | |
|---|---|
 | |
| Young woman with malar rash, typically found in systemic lupus erythematosus (SLE) | |
| Specialty | Rheumatology, immunology, gastroenterology, neurology, dermatology, endocrinology |
| Symptoms | Wide-ranging, depends on the condition. Commonly include, although by no means restricted to, low grade fever, feeling tired[1] |
| Usual onset | Adulthood[1] |
| Types | List of autoimmune diseases (alopecia areata, vitiligo, celiac disease, diabetes mellitus type 1, Hashimoto's disease, Graves' disease, inflammatory bowel disease, multiple sclerosis, psoriasis, rheumatoid arthritis, systemic lupus erythematosus, others)[1] |
| Medication | Nonsteroidal anti-inflammatory drugs, immunosuppressants, intravenous immunoglobulin[1][2] |
| Frequency | 10% (UK)[3] |
An autoimmune disease is a condition that results from an anomalous response of the adaptive immune system, wherein it mistakenly targets and attacks healthy, functioning parts of the body as if they were foreign organisms.[1] It is estimated that there are more than 80 recognized autoimmune diseases, with recent scientific evidence suggesting the existence of potentially more than 100 distinct conditions.[4][5][6] Nearly any body part can be involved.[7]
Autoimmune diseases are a separate class from autoinflammatory diseases. Both are characterized by an immune system malfunction which may cause similar symptoms, such as rash, swelling, or fatigue, but the cardinal cause or mechanism of the diseases are different. A key difference is a malfunction of the innate immune system in autoinflammatory diseases, whereas in autoimmune diseases there is a malfunction of the adaptive immune system.[8]
Symptoms of autoimmune diseases can significantly vary, primarily based on the specific type of the disease and the body part that it affects. Symptoms are often diverse and can be fleeting, fluctuating from mild to severe, and typically comprise low-grade fever, fatigue, and general malaise.[1] However, some autoimmune diseases may present with more specific symptoms such as joint pain, skin rashes (e.g., urticaria), or neurological symptoms.
The exact causes of autoimmune diseases remain unclear and are likely multifactorial, involving both genetic and environmental influences.[7] While some diseases like lupus exhibit familial aggregation, suggesting a genetic predisposition, other cases have been associated with infectious triggers or exposure to environmental factors, implying a complex interplay between genes and environment in their etiology.
Some of the most common diseases that are generally categorized as autoimmune include celiac disease, type 1 diabetes, Graves' disease, inflammatory bowel diseases (such as Crohn's disease and ulcerative colitis), multiple sclerosis, alopecia areata,[9] Addison's disease, pernicious anemia, psoriasis, rheumatoid arthritis, and systemic lupus erythematosus. Diagnosing autoimmune diseases can be challenging due to their diverse presentations and the transient nature of many symptoms.[1]
Treatment modalities for autoimmune diseases vary based on the type of disease and its severity.[1] Therapeutic approaches primarily aim to manage symptoms, reduce immune system activity, and maintain the body's ability to fight diseases. Nonsteroidal anti-inflammatory drugs (NSAIDs) and immunosuppressants are commonly used to reduce inflammation and control the overactive immune response. In certain cases, intravenous immunoglobulin may be administered to regulate the immune system.[2] Despite these treatments often leading to symptom improvement, they usually do not offer a cure and long-term management is often required.[1]
In terms of prevalence, a UK study found that 10% of the population were affected by an autoimmune disease.[3] Women are more commonly affected than men. Autoimmune diseases predominantly begin in adulthood, although they can start at any age.[1] The initial recognition of autoimmune diseases dates back to the early 1900s, and since then, advancements in understanding and management of these conditions have been substantial, though much more is needed to fully unravel their complex etiology and pathophysiology.[10]
Signs and symptoms
[edit]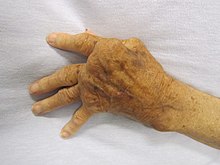
Autoimmune diseases represent a vast and diverse category of disorders that, despite their differences, share some common symptomatic threads.[1] These shared symptoms occur as a result of the body's immune system mistakenly attacking its own cells and tissues, causing inflammation and damage. However, due to the broad range of autoimmune diseases, the specific presentation of symptoms can significantly vary based on the type of disease, the organ systems affected, and individual factors such as age, sex, hormonal status, and environmental influences.[1]
An individual may simultaneously have more than one autoimmune disease (known as polyautoimmunity), further complicating the symptomatology.[1]
Common symptoms
[edit]Symptoms that are commonly associated with autoimmune diseases include:[11]
- fatigue. This is the most common complaint of people with autoimmune disease.[12] A 2015 US survey found that 98% of people with autoimmune diseases experienced fatigue, 89% said it was a "major issue", 68% said "fatigue is anything but normal. It is profound and prevents [them] from doing the simplest everyday tasks." and 59% said it was "probably the most debilitating symptom of having an AD."[13]
- low-grade fever
- malaise (a general feeling of discomfort or unease)
- muscle aches
- joint pain
- skin rashes
Specific autoimmune diseases have a wide range of other symptoms, with examples including dry mouth, dry eyes, tingling and numbness in parts of the body, unexpected weight loss or gain, and diarrhoea.
Patterns of symptom occurrence
[edit]These symptoms often reflect the body's systemic inflammatory response. However, their occurrence and intensity can fluctuate over time, leading to periods of heightened disease activity, referred to as flare-ups, and periods of relative inactivity, known as remissions.
The specific presentation of symptoms largely depends on the location and type of autoimmune response. For instance, in rheumatoid arthritis, an autoimmune disease primarily affecting the joints, symptoms typically include joint pain, swelling, and stiffness. On the other hand, type 1 diabetes, which results from an autoimmune attack on the insulin-producing cells of the pancreas, primarily presents with symptoms related to high blood sugar, such as increased thirst, frequent urination, and unexplained weight loss.
Commonly affected body areas
[edit]Commonly affected areas in autoimmune diseases include blood vessels, connective tissues, joints, muscles, red blood cells, skin, and endocrine glands such as the thyroid gland (in diseases like Hashimoto's thyroiditis and Graves' disease) and the pancreas (in type 1 diabetes). The impacts of these diseases can range from localized damage to certain tissues, alteration in organ growth and function, to more systemic effects when multiple tissues throughout the body are affected.[14]
Value of tracking symptom occurrence
[edit]The appearance of these signs and symptoms can not only provide clues for the diagnosis of an autoimmune condition, often in conjunction with tests for specific biological markers, but also help monitor disease progression and response to treatment.[15] Ultimately, due to the diverse nature of autoimmune diseases, a multidimensional approach is often needed for the management of these conditions, taking into consideration the variety of symptoms and their impacts on individuals' lives.
Types
[edit]While it is estimated that over 80 recognized types of autoimmune diseases exist, this section provides an overview of some of the most common and well-studied forms.[1][16][17]
Coeliac disease
[edit]Coeliac disease is an immune reaction to eating gluten, a protein found in wheat, barley, and rye.[18] For those with the disease, eating gluten triggers an immune response in the small intestine, leading to damage on the villi, small fingerlike projections that line the small intestine and promote nutrient absorption.[18] This explains the increased risk of gastrointestinal cancers, as the gastrointestinal tract includes the esophagus, stomach, small intestine, large intestine, rectum, and anus, all areas that the ingested gluten would traverse in digestion.[18] The incidence of gastrointestinal cancer can be partially reduced or eliminated if a patient removes gluten from their diet.[18][19][20][21][22] Additionally, coeliac disease is correlated with lymphoproliferative disorders.[18]
Graves' disease
[edit]Graves' disease is a condition characterized by development of autoantibodies to thyroid-stimulating hormone (TSH) receptors (TRAb). The binding of TRAb autoantibodies to TSH receptor results in unregulated production and release of thyroid hormone,[23] which can lead to stimulatory effects such as rapid heart rate, weight loss, nervousness, and irritability. Other symptoms more specific to Graves' disease include bulging eyes and swelling of the lower legs.
Inflammatory bowel disease
[edit]Inflammatory bowel disease (IBD) encompasses conditions characterized by chronic inflammation of the digestive tract, including Crohn's disease and ulcerative colitis. In both cases, individuals with IBD lose immune tolerance for normal bacteria present in the gut microbiome.[18] Symptoms include severe diarrhea, abdominal pain, fatigue, and weight loss. IBD is associated with cancers of the gastrointestinal tract and some lymphoproliferative cancers.[18]
Multiple sclerosis
[edit]Multiple sclerosis (MS) is a neurodegenerative disease in which the immune system attacks myelin, a protective covering of nerve fibers in the central nervous system, causing communication problems between the brain and the rest of the body. Symptoms can include fatigue, difficulty walking, numbness or tingling, muscle weakness, and problems with coordination and balance.[24] MS is associated with an increased risk of central nervous system cancer, primarily in the brain.[18]
Rheumatoid arthritis
[edit]Rheumatoid arthritis (RA) primarily targets the joints, causing persistent inflammation that results in joint damage and pain. It is often symmetrical, meaning that if one hand or knee has it, the other one does too. RA can also affect the heart, lungs, and eyes. Additionally, the chronic inflammation and over-activation of the immune system creates an environment that favors further malignant transformation of other cells, perhaps explaining the associations with cancer of the lungs and skin as well as the increased risk of other hematologic cancers, none of which are directly affected by the inflammation of joints.[25][26]
Psoriasis and psoriatic arthritis
[edit]Psoriasis is a skin condition characterized by the rapid buildup of skin cells, leading to scaling on the skin's surface. Inflammation and redness around the scales is common.[27] Some individuals with psoriasis also develop psoriatic arthritis, which causes joint pain, stiffness, and swelling.[28]
Sjögren's syndrome
[edit]Sjögren syndrome (SjS, SS) is a long-term autoimmune disease that affects the body's moisture-producing glands (lacrimal and salivary),[29] and often seriously affects other organ systems, such as the lungs, kidneys, and nervous system.
Systemic lupus erythematosus
[edit]Systemic lupus erythematosus (SLE), referred to simply as lupus, is a systemic autoimmune disease that affects multiple organs, including the skin, joints, kidneys, and the nervous system. It is characterized by a widespread loss of immune tolerance.[30] The disease is characterized by periods of flares and remissions, and symptoms range from mild to severe. Women, especially those of childbearing age, are disproportionately affected.[31]
Type 1 diabetes
[edit]Type 1 diabetes is a condition resulting from the immune system attacking insulin-producing beta cells in the pancreas, leading to high blood sugar levels. Symptoms include increased thirst, frequent urination, and unexplained weight loss. It is most commonly diagnosed in children and young adults.[32]
Undifferentiated connective tissue disease
[edit]Undifferentiated connective tissue disease (UCTD) occurs when people have features of connective tissue disease, such as blood test results and external characteristics, but do not fulfill the diagnostic criteria established for any one connective tissue disease. Some 30–40% transition to a specific connective tissue disease over time.
Causes
[edit]The exact causes of autoimmune diseases remain largely unknown;[7] however, research has suggested that a combination of genetic, environmental, and hormonal factors, as well as certain infections, may contribute to the development of these disorders.[1]
The human immune system is equipped with several mechanisms to maintain a delicate balance between defending against foreign invaders and protecting its own cells. To achieve this, it generates both T cells and B cells, which are capable of reacting with self-proteins. However, in a healthy immune response, self-reactive cells are generally either eliminated before they become active, rendered inert via a process called anergy, or their activities are suppressed by regulatory cells.
Genetics
[edit]A familial tendency to develop autoimmune diseases suggests a genetic component. Some conditions, like lupus and multiple sclerosis, often occur in several members of the same family, indicating a potential hereditary link. Additionally, certain genes have been identified that increase the risk of developing specific autoimmune diseases.
Genetic predisposition
[edit]Evidence suggests a strong genetic component in the development of autoimmune diseases.[33] For instance, conditions such as lupus and multiple sclerosis frequently appear in multiple members of the same family, signifying a potential hereditary link. Furthermore, certain genes have been identified that augment the risk of developing specific autoimmune diseases.[34]
Experimental methods like genome-wide association studies (GWAS) have proven instrumental in pinpointing genetic risk variants potentially responsible for autoimmune diseases. For example, these studies have been used to identify risk variants for diseases such as type 1 diabetes and rheumatoid arthritis.[35]
In twin studies, autoimmune diseases consistently demonstrate a higher concordance rate among identical twins compared with fraternal twins. For instance, the rate in multiple sclerosis is 35% in identical twins compared to 6% in fraternal twins.[36][37]
Balancing infection and autoimmunity
[edit]There is increasing evidence that certain genes selected during evolution offer a balance between susceptibility to infection and the capacity to avoid autoimmune diseases.[38] For example, variants in the ERAP2 gene provide some resistance to infection even though they increase the risk of autoimmunity (positive selection). In contrast, variants in the TYK2 gene protect against autoimmune diseases but increase the risk of infection (negative selection). This suggests the benefits of infection resistance may outweigh the risks of autoimmune diseases, particularly given the historically high risk of infection.[38]
Several experimental methods such as the genome-wide association studies (GWAS) have been used to identify genetic risk variants that may be responsible[39] for diseases such as type 1 diabetes and rheumatoid arthritis.[40]
Environmental factors
[edit]A significant number of environmental factors have been implicated in the development and progression of various autoimmune diseases, either directly or as catalysts. Current research suggests that up to seventy percent of autoimmune diseases could be attributed to environmental influences, which encompass an array of elements such as chemicals, infectious agents, dietary habits, and gut dysbiosis. However, a unifying theory that definitively explains the onset of autoimmune diseases remains elusive, emphasizing the complexity and multifaceted nature of these conditions.[41]
Various environmental triggers are identified, some of which include:
- Impaired oral tolerance
- Gut dysbiosis
- Increased gut permeability
- Heightened immune reactivity
Chemicals, which are either a part of the immediate environment or found in drugs, are key players in this context. Examples of such chemicals include hydrazines, hair dyes, trichloroethylene, tartrazines, hazardous wastes, and industrial emissions.[42]
Ultraviolet (UV) radiation has been implicated as a potential causative factor in the development of autoimmune diseases, such as dermatomyositis.[43] Furthermore, exposure to pesticides has been linked with an increased risk of developing rheumatoid arthritis.[44] Vitamin D, on the other hand, appears to play a protective role, particularly in older populations, by preventing immune dysfunctions.[45]
Infectious agents are also being increasingly recognized for their role as T cell activators — a crucial step in triggering autoimmune diseases. The exact mechanisms by which they contribute to disease onset remain to be fully understood. For instance, certain autoimmune conditions like Guillain-Barre syndrome and rheumatic fever are thought to be triggered by infections.[46] Furthermore, analysis of large-scale data has revealed a significant link between SARS-CoV-2 infection (the causative agent of COVID-19) and an increased risk of developing a wide range of new-onset autoimmune diseases.[47]
Sex
[edit]Women typically make up some 80% of autoimmune disease patients.[48] Whilst many proposals have been made for the cause of this high weighting, no clear explanation is available.[49][50] A possible role for hormonal factors has been suggested.[51] For example, some autoimmune diseases tend to flare during pregnancy (possibly as an evolutionary mechanism to increase health protection for the child),[50] when hormone levels are high, and improve after menopause, when hormone levels decrease. Women may also naturally have autoimmune disease trigger events in puberty and pregnancy.[48] Under-reporting by men may also be a factor, as men may interact less with the health system than women.[52][53][54][55][56]
Infections
[edit]Certain viral and bacterial infections have been linked to autoimmune diseases.[57] For instance, research suggests that the bacterium that causes strep throat, Streptococcus pyogenes, might trigger rheumatic fever, an autoimmune response affecting the heart.[58] Similarly, some studies propose a link between the Epstein–Barr virus, responsible for mononucleosis, and the subsequent development of multiple sclerosis or lupus.
Dysregulated immune response
[edit]Another area of interest is the immune system's ability to distinguish between self and non-self, a function that is compromised in autoimmune diseases. In healthy individuals, immune tolerance prevents the immune system from attacking the body's own cells. When this process fails, the immune system may produce antibodies against its own tissues, leading to an autoimmune response.[59]
Negative selection and the role of the thymus
[edit]The elimination of self-reactive T cells occurs primarily through a mechanism known as "negative selection" within the thymus, an organ responsible for the maturation of T cells.[60] This process serves as a key line of defense against autoimmunity. If these protective mechanisms fail, a pool of self-reactive cells can become functional within the immune system, contributing to the development of autoimmune diseases.
Molecular mimicry
[edit]Some infectious agents, like Campylobacter jejuni, bear antigens that resemble, but are not identical to, the body's self-molecules. This phenomenon, known as molecular mimicry, can lead to cross-reactivity, where the immune response to such infections inadvertently results in the production of antibodies that also react with self-antigens.[61] An example of this is Guillain–Barré syndrome, in which antibodies generated in response to a C. jejuni infection also react with the gangliosides in the myelin sheath of peripheral nerve axons.[62]
Diagnosis
[edit]Diagnosing autoimmune disorders can be complex due to the wide range of diseases within this category and their often overlapping symptoms. Accurate diagnosis is crucial for determining appropriate treatment strategies. Generally, the diagnostic process involves a combination of medical history evaluation, physical examination, laboratory tests, and, in some cases, imaging or biopsies.[63]
Medical history and examination
[edit]The first step in diagnosing autoimmune disorders typically involves a thorough evaluation of the patient's medical history and a comprehensive physical examination.[34] Clinicians often pay close attention to the patient's symptoms, family history of autoimmune diseases, and any exposure to environmental factors that might trigger an autoimmune response. The physical examination can reveal signs of inflammation or organ damage, which are common features of autoimmune disorders.
Laboratory tests
[edit]Laboratory testing plays a pivotal role in the diagnosis of autoimmune diseases. These tests can identify the presence of certain autoantibodies or other immune markers that indicate a self-directed immune response.
- Autoantibody testing: Many autoimmune diseases are characterized by the presence of autoantibodies. Blood tests can identify these antibodies, which are directed against the body's own tissues.[64] For example, antinuclear antibody (ANA) testing is commonly used in the diagnosis of systemic lupus erythematosus (SLE) and other autoimmune diseases.
- Complete Blood Count (CBC): A CBC can provide valuable information about the number and characteristics of different blood cells, which can be affected in some autoimmune diseases.[65][42][63]
- C-Reactive Protein (CRP) and Erythrocyte Sedimentation Rate (ESR): These tests measure the levels of inflammation in the body, which is often elevated in autoimmune disorders.[63][42]
- Organ-specific tests: Certain autoimmune diseases target specific organs, so tests to evaluate the function of these organs can aid in diagnosis. For example, thyroid function tests are used in diagnosing autoimmune thyroid disorders, while a biopsy can diagnose celiac disease by identifying damage to the small intestine.
Imaging studies
[edit]In some cases, imaging studies may be used to assess the extent of organ involvement and damage. For example, chest x-rays or CT scans can identify lung involvement in diseases like rheumatoid arthritis or systemic lupus erythematosus, while an MRI can reveal inflammation or damage in the brain and spinal cord in multiple sclerosis.
Differential diagnosis
[edit]Given the variety and nonspecific nature of symptoms that can be associated with autoimmune diseases, differential diagnosis—determining which of several diseases with similar symptoms is causing a patient's illness—is an important part of the diagnostic process. This often involves ruling out other potential causes of symptoms, such as infections, malignancies, or genetic disorders.
Multidisciplinary approach
[edit]Given the systemic nature of many autoimmune disorders, a multidisciplinary approach may be necessary for their diagnosis and management. This can involve rheumatologists, endocrinologists, gastroenterologists, neurologists, dermatologists, and other specialists, depending on the organs or systems affected by the disease.
In summary, the diagnosis of autoimmune disorders is a complex process that requires a thorough evaluation of clinical, laboratory, and imaging data. Due to the diverse nature of these diseases, an individualized approach, often involving multiple specialists, is crucial for an accurate diagnosis.
Treatment
[edit]Treatment depends on the type and severity of the condition. The majority of the autoimmune diseases are chronic and there is no definitive cure, but symptoms can be alleviated and controlled with treatment.[11] Standard treatment methods include:[11]
- Vitamin or hormone supplements for what the body is lacking due to the disease (insulin, vitamin B12, thyroid hormone, etc.)
- Blood transfusions if the disease is blood related
- Physical therapy if the disease impacts bones, joints, or muscles
Traditional treatment options include immunosuppressant drugs to reduce the immune response against the body's own tissues, such as:[66]
- Non-steroidal anti-inflammatory drugs (NSAIDs) to reduce inflammation
- Glucocorticoids to reduce inflammation
- Disease-modifying anti-rheumatic drugs (DMARDs) to decrease the damaging tissue and organ effects of the inflammatory autoimmune response
Because immunosuppressants weaken the overall immune response, relief of symptoms must be balanced with preserving the patient's ability to combat infections, which could potentially be life-threatening.[67]
Non-traditional treatments are being researched, developed, and used, especially when traditional treatments fail. These methods aim to either block the activation of pathogenic cells in the body, or alter the pathway that suppresses these cells naturally.[67][68] These treatments aim to be less toxic to the patient and have more specific targets.[68] Such options include:
- Monoclonal antibodies that can be used to block pro-inflammatory cytokines
- Antigen-specific immunotherapy which allows immune cells to specifically target the abnormal cells that cause autoimmune disease[68]
- Co-stimulatory blockade that works to block the pathway that leads to the autoimmune response
- Regulatory T cell therapy that utilizes this special type of T cell to suppress the autoimmune response[67]
- Thymoquinone, a compound found the flower Nigella sativa, has been studied for potential in treating several autoimmune diseases due to its effects on inflammation.[69][70]
Manage Autoimmune Disorders with Yoga:[71] Yoga, an ancient practice that integrates physical postures, breath control, and meditation, has shown promise in managing autoimmune disorders. Research indicates that regular yoga practice can reduce stress, a known trigger for autoimmune flare-ups, by promoting relaxation and mental clarity.
Epidemiology
[edit]The first estimate of US prevalence for autoimmune diseases as a group was published in 1997 by Jacobson, et al. They reported US prevalence to be around 9 million, applying prevalence estimates for 24 diseases to a US population of 279 million.[72] Jacobson's work was updated by Hayter & Cook in 2012.[73] This study used Witebsky's postulates, as revised by Rose & Bona,[74] to extend the list to 81 diseases and estimated overall cumulative US prevalence for the 81 autoimmune diseases at 5.0%, with 3.0% for males and 7.1% for females.
The estimated community prevalence, which takes into account the observation that many people have more than one autoimmune disease, was 4.5% overall, with 2.7% for males and 6.4% for females.[73] National Health and Nutrition Examination Surveys conducted in the US from the 1980s to 2020 showed an increase of antinuclear antibodies, a common biomarker for autoimmune diseases. This shows that there has been an increase in the prevalence of autoimmune diseases pointing to a stronger influence of environment factors as a risk factor for autoimmune diseases.[75]
A 2024 estimate was that 1 in 15 people in the U.S. had at least one autoimmune disease.[76]
Research
[edit]In both autoimmune and inflammatory diseases, the condition arises through aberrant reactions of the human adaptive or innate immune systems. In autoimmunity, the patient's immune system is activated against the body's own proteins. In chronic inflammatory diseases, neutrophils and other leukocytes are constitutively recruited by cytokines and chemokines, resulting in tissue damage.[77]
Mitigation of inflammation by activation of anti-inflammatory genes and the suppression of inflammatory genes in immune cells is a promising therapeutic approach.[78][79][80] There is a body of evidence that once the production of autoantibodies has been initialized, autoantibodies have the capacity to maintain their own production.[81]
Stem-cell therapy
[edit]Stem cell transplantation is being studied and has shown promising results in certain cases.[82]
Medical trials to replace the pancreatic β cells that are destroyed in type 1 diabetes are in progress.[83]
Altered glycan theory
[edit]According to this theory, the effector function of the immune response is mediated by the glycans (polysaccharides) displayed by the cells and humoral components of the immune system. Individuals with autoimmunity have alterations in their glycosylation profile such that a proinflammatory immune response is favored. It is further hypothesized that individual autoimmune diseases will have unique glycan signatures.[84]
Hygiene hypothesis
[edit]According to the hygiene hypothesis, high levels of cleanliness expose children to fewer antigens than in the past, causing their immune systems to become overactive and more likely to misidentify own tissues as foreign, resulting in autoimmune or allergic conditions such as asthma.[85]
Vitamin D influence on immune response
[edit]Vitamin D is known as an immune regulator that assists in the adaptive and innate immune response.[86][87] A deficiency in vitamin D, from hereditary or environmental influence, can lead to a more inefficient and weaker immune response and seen as a contributing factor to the development of autoimmune diseases.[87] With vitamin D present, vitamin D response elements (VDRE) are encoded and expressed via pattern recognition receptors (PRR) responses and the genes associated with those responses.[86] The specific DNA target sequence expressed is known as 1,25-(OH)2D3.[86] The expression of 1,25-(OH)2D3 can be induced by macrophages, dendritic cells, T-cells, and B-cells.[86] In the presence of 1,25-(OH)2D3, the immune system's production of inflammatory cytokines are suppressed and more tolerogenic regulatory T-cells are expressed.[86] This is due to vitamin D's influence on cell maturation, specifically T-cells, and their phenotype expression.[86] Lack of 1,25-(OH)2D3 expression can lead to less tolerant regulatory T-cells, larger presentation of antigens to less tolerant T-cells, and increased inflammatory response.[86]
See also
[edit]References
[edit]- ^ a b c d e f g h i j k l m n o "Autoimmune diseases". Office on Women's Health. U.S. Department of Health and Human Services. 22 February 2021. Archived from the original on 5 October 2016. Retrieved 5 October 2016.
- ^ a b Katz U, Shoenfeld Y, Zandman-Goddard G (2011). "Update on intravenous immunoglobulins (IVIg) mechanisms of action and off- label use in autoimmune diseases". Current Pharmaceutical Design. 17 (29): 3166–3175. doi:10.2174/138161211798157540. PMID 21864262.
- ^ a b Conrad N, Misra S, Verbakel JY, Verbeke G, Molenberghs G, Taylor PN, et al. (June 2023). "Incidence, prevalence, and co-occurrence of autoimmune disorders over time and by age, sex, and socioeconomic status: a population-based cohort study of 22 million individuals in the UK". Lancet. 401 (10391): 1878–1890. doi:10.1016/S0140-6736(23)00457-9. PMID 37156255. S2CID 258529606.
- ^ "List of Autoimmune Diseases". Autoimmune Registry Inc. Retrieved 2022-06-06.
- ^ Angum F, Khan T, Kaler J, Siddiqui L, Hussain A (May 2020). "The Prevalence of Autoimmune Disorders in Women: A Narrative Review". Cureus. 12 (5): e8094. doi:10.7759/cureus.8094. PMC 7292717. PMID 32542149. S2CID 219447364.
- ^ "Assessment of NIH Research on Autoimmune Diseases". www.nationalacademies.org. Retrieved 2022-06-13.
- ^ a b c Borgelt LM (2010). Women's Health Across the Lifespan: A Pharmacotherapeutic Approach. ASHP. p. 579. ISBN 978-1-58528-194-7. Archived from the original on 2017-09-08.
- ^ Zen M, Gatto M, Domeneghetti M, Palma L, Borella E, Iaccarino L, et al. (October 2013). "Clinical guidelines and definitions of autoinflammatory diseases: contrasts and comparisons with autoimmunity-a comprehensive review". Clinical Reviews in Allergy & Immunology. 45 (2): 227–235. doi:10.1007/s12016-013-8355-1. PMID 23322404. S2CID 23061331.
- ^ Erjavec SO, Gelfman S, Abdelaziz AR, Lee EY, Monga I, Alkelai A, et al. (February 2022). "Whole exome sequencing in Alopecia Areata identifies rare variants in KRT82". Nature Communications. 13 (1): 800. Bibcode:2022NatCo..13..800E. doi:10.1038/s41467-022-28343-3. PMC 8831607. PMID 35145093.
- ^ Ananthanarayan R, Paniker CK (2005). Ananthanarayan and Paniker's Textbook of Microbiology. Orient Blackswan. p. 169. ISBN 978-81-250-2808-6. Archived from the original on 2017-09-08.
- ^ a b c "Autoimmune disorders". MedlinePlus, US National Library of Medicine. 16 July 2013. Retrieved 12 July 2023.
- ^ Zielinski MR, Systrom DM, Rose NR (2019-08-06). "Fatigue, Sleep, and Autoimmune and Related Disorders". Frontiers in Immunology. 10: 1827. doi:10.3389/fimmu.2019.01827. PMC 6691096. PMID 31447842.
- ^ Baumer K (2015-03-23). "Fatigue Survey Results Released". Autoimmune Association. Retrieved 2024-01-25.
- ^ Smith DA, Germolec DR (October 1999). "Introduction to immunology and autoimmunity". Environmental Health Perspectives. 107 (Suppl 5): 661–665. doi:10.1289/ehp.99107s5661. PMC 1566249. PMID 10502528.
- ^ National Research Council (US) Subcommittee on Immunotoxicology (1992). Autoimmune Diseases. National Academies Press (US).
- ^ "Autoimmune Disease List • AARDA". AARDA. 2016-06-01. Retrieved 2019-03-21.
- ^ Hohlfeld R, Dornmair K, Meinl E, Wekerle H (February 2016). "The search for the target antigens of multiple sclerosis, part 1: autoreactive CD4+ T lymphocytes as pathogenic effectors and therapeutic targets". The Lancet. Neurology. 15 (2): 198–209. doi:10.1016/S1474-4422(15)00334-8. PMID 26724103. S2CID 20082472.
- ^ a b c d e f g h Franks AL, Slansky JE (April 2012). "Multiple associations between a broad spectrum of autoimmune diseases, chronic inflammatory diseases and cancer". Anticancer Research. 32 (4): 1119–1136. PMC 3349285. PMID 22493341.
- ^ Meresse B, Ripoche J, Heyman M, Cerf-Bensussan N (January 2009). "Celiac disease: from oral tolerance to intestinal inflammation, autoimmunity and lymphomagenesis". Mucosal Immunology. 2 (1): 8–23. doi:10.1038/mi.2008.75. PMID 19079330. S2CID 24980464.
- ^ Green PH, Fleischauer AT, Bhagat G, Goyal R, Jabri B, Neugut AI (August 2003). "Risk of malignancy in patients with celiac disease". The American Journal of Medicine. 115 (3): 191–195. doi:10.1016/s0002-9343(03)00302-4. PMID 12935825.
- ^ Volta U, Vincentini O, Silano M (2011). "Papillary cancer of thyroid in celiac disease". Journal of Clinical Gastroenterology. 45 (5): e44–e46. doi:10.1097/mcg.0b013e3181ea11cb. PMID 20697293. S2CID 24754769.
- ^ Catassi C, Bearzi I, Holmes GK (April 2005). "Association of celiac disease and intestinal lymphomas and other cancers". Gastroenterology. 128 (4 Suppl 1): S79–S86. doi:10.1053/j.gastro.2005.02.027. PMID 15825131.
- ^ "Graves disease: MedlinePlus Genetics". MedlinePlus. 2013-07-01. Retrieved 2023-06-25.
- ^ Frohman EM, Racke MK, Raine CS (March 2006). "Multiple sclerosis--the plaque and its pathogenesis". The New England Journal of Medicine. 354 (9): 942–955. doi:10.1056/nejmra052130. PMID 16510748.
- ^ Turesson C, Matteson EL (2009). "Clinical Features of Rheumatoid Arthritis: Extra-Articular Manifestations". Rheumatoid Arthritis. Elsevier. pp. 62–67. doi:10.1016/b978-032305475-1.50014-8. ISBN 978-0-323-05475-1.
- ^ Khurana R, Berney SM (October 2005). "Clinical aspects of rheumatoid arthritis". Pathophysiology. 12 (3): 153–165. doi:10.1016/j.pathophys.2005.07.009. PMID 16125918.
- ^ "Psoriasis - Psoriatic Arthritis". National Library of Medicine. 2019-02-20. Retrieved 2023-06-25.
- ^ "Psoriatic Arthritis". National Library of Medicine. 2016-08-25. Retrieved 2023-06-25.
- ^ "What Is Sjögren's Syndrome? Fast Facts". NIAMS. November 2014. Archived from the original on 4 July 2016. Retrieved 15 July 2016.
- ^ Tsokos GC (December 2011). "Systemic lupus erythematosus". The New England Journal of Medicine. 365 (22): 2110–2121. doi:10.1056/nejmra1100359. PMID 22129255.
- ^ "Lupus". Office on Women's Health. 2021-02-18. Retrieved 2023-06-25.
- ^ "Type 1 Diabetes - Juvenile Diabetes". National Library of Medicine. 2020-06-29. Retrieved 2023-06-25.
- ^ Antonelli A, Ferrari SM, Ragusa F, Elia G, Paparo SR, Ruffilli I, et al. (January 2020). "Graves' disease: Epidemiology, genetic and environmental risk factors and viruses". Best Practice & Research. Clinical Endocrinology & Metabolism. 34 (1). Elsevier BV: 101387. doi:10.1016/j.beem.2020.101387. PMID 32107168. S2CID 211555101.
- ^ a b Wang L, Wang FS, Gershwin ME (October 2015). "Human autoimmune diseases: a comprehensive update". Journal of Internal Medicine. 278 (4). Wiley: 369–395. doi:10.1111/joim.12395. PMID 26212387. S2CID 24386085.
- ^ Lettre G, Rioux JD (October 2008). "Autoimmune diseases: insights from genome-wide association studies". Human Molecular Genetics. 17 (R2). Oxford University Press (OUP): R116–R121. doi:10.1093/hmg/ddn246. PMC 2782355. PMID 18852199.
- ^ Willer CJ, Dyment DA, Risch NJ, Sadovnick AD, Ebers GC (October 2003). "Twin concordance and sibling recurrence rates in multiple sclerosis". Proceedings of the National Academy of Sciences of the United States of America. 100 (22): 12877–12882. Bibcode:2003PNAS..10012877W. doi:10.1073/pnas.1932604100. PMC 240712. PMID 14569025.
- ^ Filippi M, Bar-Or A, Piehl F, Preziosa P, Solari A, Vukusic S, Rocca MA (November 2018). "Multiple sclerosis". Nature Reviews. Disease Primers. 4 (1): 43. doi:10.1038/s41572-018-0041-4. PMID 30410033. S2CID 53238233.
- ^ a b McKinney EF, Lee JC, Jayne DR, Lyons PA, Smith KG (July 2015). "T-cell exhaustion, co-stimulation and clinical outcome in autoimmunity and infection". Nature. 523 (7562). Springer Science and Business Media LLC: 612–616. Bibcode:2015Natur.523..612M. doi:10.1038/nature14468. PMC 4623162. PMID 26123020.
- ^ Gregersen PK, Olsson LM (2009). "Recent advances in the genetics of autoimmune disease". Annual Review of Immunology. 27: 363–391. doi:10.1146/annurev.immunol.021908.132653. PMC 2992886. PMID 19302045.
- ^ Hill NJ, King C, Flodstrom-Tullberg M (May 2008). "Recent acquisitions on the genetic basis of autoimmune disease". Frontiers in Bioscience. 13 (13): 4838–4851. doi:10.2741/3043. PMID 18508549.
- ^ Vojdani A (2014). "A Potential Link between Environmental Triggers and Autoimmunity". Autoimmune Diseases. 2014: 437231. doi:10.1155/2014/437231. PMC 3945069. PMID 24688790.
- ^ a b c "Autoimmune Diseases: Symptoms & Causes". Boston Children's Hospital. Archived from the original on 7 April 2021. Retrieved 23 March 2020.
- ^ Shah M, Targoff IN, Rice MM, Miller FW, Rider LG (July 2013). "Brief report: ultraviolet radiation exposure is associated with clinical and autoantibody phenotypes in juvenile myositis". Arthritis and Rheumatism. 65 (7): 1934–1941. doi:10.1002/art.37985. PMC 3727975. PMID 23658122.
- ^ Meyer A, Sandler DP, Beane Freeman LE, Hofmann JN, Parks CG (July 2017). "Pesticide Exposure and Risk of Rheumatoid Arthritis among Licensed Male Pesticide Applicators in the Agricultural Health Study". Environmental Health Perspectives. 125 (7): 077010. doi:10.1289/EHP1013. PMC 5744649. PMID 28718769.
- ^ Meier HC, Sandler DP, Simonsick EM, Parks CG (December 2016). "Association between Vitamin D Deficiency and Antinuclear Antibodies in Middle-Aged and Older U.S. Adults". Cancer Epidemiology, Biomarkers & Prevention. 25 (12): 1559–1563. doi:10.1158/1055-9965.EPI-16-0339. PMC 5135624. PMID 27543618.
- ^ Wucherpfennig KW (October 2001). "Mechanisms for the induction of autoimmunity by infectious agents". The Journal of Clinical Investigation. 108 (8): 1097–1104. doi:10.1172/JCI14235. PMC 209539. PMID 11602615.
- ^ Sharma C, Bayry J (July 2023). "High risk of autoimmune diseases after COVID-19". Nature Reviews. Rheumatology. 19 (7): 399–400. doi:10.1038/s41584-023-00964-y. PMC 10096101. PMID 37046064.
- ^ a b Angum F, Khan T, Kaler J, Siddiqui L, Hussain A (May 2020). "The Prevalence of Autoimmune Disorders in Women: A Narrative Review". Cureus. 12 (5): e8094. doi:10.7759/cureus.8094. PMC 7292717. PMID 32542149.
- ^ Kronzer VL, Bridges SL, Davis JM (March 2021). "Why women have more autoimmune diseases than men: An evolutionary perspective". Evolutionary Applications. 14 (3): 629–633. Bibcode:2021EvApp..14..629K. doi:10.1111/eva.13167. PMC 7980266. PMID 33767739.
- ^ a b Moyer MW (September 1, 2021). "Why Nearly 80 Percent of Autoimmune Sufferers Are Female". Scientific American.
- ^ Moulton VR (2018-10-04). "Sex Hormones in Acquired Immunity and Autoimmune Disease". Frontiers in Immunology. 9. Frontiers Media SA: 2279. doi:10.3389/fimmu.2018.02279. PMC 6180207. PMID 30337927.
- ^ Schlichthorst M, Sanci LA, Pirkis J, Spittal MJ, Hocking JS (October 2016). "Why do men go to the doctor? Socio-demographic and lifestyle factors associated with healthcare utilisation among a cohort of Australian men". BMC Public Health. 16 (Suppl 3): 1028. doi:10.1186/s12889-016-3706-5. PMC 5103250. PMID 28185596.
- ^ "Women Visit Doctor More Often than Men". NCHS Pressroom. Centers for Disease Control and Prevention. July 26, 2001. Retrieved 2024-01-25.
- ^ Billock J (22 May 2018). "Pain bias: The health inequality rarely discussed". BBC. Retrieved 2024-01-25.
- ^ Hunt K, Adamson J, Hewitt C, Nazareth I (April 2011). "Do women consult more than men? A review of gender and consultation for back pain and headache". Journal of Health Services Research & Policy. 16 (2): 108–117. doi:10.1258/jhsrp.2010.009131. PMC 3104816. PMID 20819913.
- ^ Wang Y, Hunt K, Nazareth I, Freemantle N, Petersen I (August 2013). "Do men consult less than women? An analysis of routinely collected UK general practice data". BMJ Open. 3 (8): e003320. doi:10.1136/bmjopen-2013-003320. PMC 3753483. PMID 23959757.
- ^ Getts DR, Chastain EM, Terry RL, Miller SD (September 2013). "Virus infection, antiviral immunity, and autoimmunity". Immunological Reviews. 255 (1). Wiley: 197–209. doi:10.1111/imr.12091. PMC 3971377. PMID 23947356.
- ^ Karthikeyan G, Guilherme L (July 2018). "Acute rheumatic fever". Lancet. 392 (10142). Elsevier BV: 161–174. doi:10.1016/s0140-6736(18)30999-1. PMID 30025809. S2CID 51702921.
- ^ Conigliaro P, Triggianese P, Ballanti E, Perricone C, Perricone R, Chimenti MS (September 2019). "Complement, infection, and autoimmunity". Current Opinion in Rheumatology. 31 (5). Ovid Technologies (Wolters Kluwer Health): 532–541. doi:10.1097/bor.0000000000000633. hdl:2108/225709. PMID 31192812. S2CID 189813866.
- ^ Bruserud Ø, Oftedal BE, Wolff AB, Husebye ES (December 2016). "AIRE-mutations and autoimmune disease". Current Opinion in Immunology. 43. Elsevier BV: 8–15. doi:10.1016/j.coi.2016.07.003. PMID 27504588.
- ^ Rojas M, Restrepo-Jiménez P, Monsalve DM, Pacheco Y, Acosta-Ampudia Y, Ramírez-Santana C, et al. (December 2018). "Molecular mimicry and autoimmunity". Journal of Autoimmunity. 95. Elsevier BV: 100–123. doi:10.1016/j.jaut.2018.10.012. PMID 30509385. S2CID 54500571.
- ^ Jasti AK, Selmi C, Sarmiento-Monroy JC, Vega DA, Anaya JM, Gershwin ME (November 2016). "Guillain-Barré syndrome: causes, immunopathogenic mechanisms and treatment". Expert Review of Clinical Immunology. 12 (11). Informa UK Limited: 1175–1189. doi:10.1080/1744666x.2016.1193006. hdl:2434/424557. PMID 27292311. S2CID 45391831.
- ^ a b c "Autoimmune disorders". MedlinePlus Medical Encyclopedia. U.S. National Library of Medicine. Retrieved 23 March 2020.
- ^ "Antinuclear Antibody (ANA)". labtestsonline.org. Retrieved 14 April 2020.
- ^ "Complete blood count (CBC)". www.mayoclinic.org. 19 December 2018. Retrieved 14 April 2020.
- ^ Li P, Zheng Y, Chen X (2017). "Drugs for Autoimmune Inflammatory Diseases: From Small Molecule Compounds to Anti-TNF Biologics". Frontiers in Pharmacology. 8: 460. doi:10.3389/fphar.2017.00460. PMC 5506195. PMID 28785220.
- ^ a b c Rosenblum MD, Gratz IK, Paw JS, Abbas AK (March 2012). "Treating human autoimmunity: current practice and future prospects". Science Translational Medicine. 4 (125): 125sr1. doi:10.1126/scitranslmed.3003504. PMC 4061980. PMID 22422994.
- ^ a b c Smilek DE, Ehlers MR, Nepom GT (May 2014). "Restoring the balance: immunotherapeutic combinations for autoimmune disease". Disease Models & Mechanisms. 7 (5): 503–513. doi:10.1242/dmm.015099. PMC 4007402. PMID 24795433.
- ^ Ali MY, Akter Z, Mei Z, Zheng M, Tania M, Khan MA (February 2021). "Thymoquinone in autoimmune diseases: Therapeutic potential and molecular mechanisms". Biomedicine & Pharmacotherapy. 134: 111157. doi:10.1016/j.biopha.2020.111157. PMID 33370631. S2CID 229714190.
- ^ Umar S, Hedaya O, Singh AK, Ahmed S (September 2015). "Thymoquinone inhibits TNF-α-induced inflammation and cell adhesion in rheumatoid arthritis synovial fibroblasts by ASK1 regulation". Toxicology and Applied Pharmacology. 287 (3): 299–305. doi:10.1016/j.taap.2015.06.017. PMC 4549173. PMID 26134265.
- ^ "Manage Auto Immune Disorder with Yoga". Kaivalyadhama. Retrieved 2024-06-26.
- ^ Jacobson DL, Gange SJ, Rose NR, Graham NM (September 1997). "Epidemiology and estimated population burden of selected autoimmune diseases in the United States". Clinical Immunology and Immunopathology. 84 (3): 223–243. doi:10.1006/clin.1997.4412. PMID 9281381.
- ^ a b Hayter SM, Cook MC (August 2012). "Updated assessment of the prevalence, spectrum and case definition of autoimmune disease". Autoimmunity Reviews. 11 (10): 754–765. doi:10.1016/j.autrev.2012.02.001. PMID 22387972.
- ^ Rose NR, Bona C (September 1993). "Defining criteria for autoimmune diseases (Witebsky's postulates revisited)". Immunology Today. 14 (9): 426–430. doi:10.1016/0167-5699(93)90244-F. PMID 8216719.
- ^ Dinse GE, Parks CG, Weinberg CR, Co CA, Wilkerson J, Zeldin DC, et al. (June 2020). "Increasing Prevalence of Antinuclear Antibodies in the United States". Arthritis & Rheumatology. 72 (6): 1026–1035. doi:10.1002/art.41214. PMC 7255943. PMID 32266792. (Retracted, see doi:10.1002/art.41214)
- ^ "Autoimmune Diseases: Causes, Symptoms, What is It & Treatment".
- ^ De Oliveira S, Rosowski EE, Huttenlocher A (2016). "Neutrophil migration in infection and wound repair: going forward in reverse". Nature Reviews. Immunology. 16 (6): 378–391. doi:10.1038/nri.2016.49. PMC 5367630. PMID 27231052.
- ^ Mukundan L, Odegaard JI, Morel CR, Heredia JE, Mwangi JW, Ricardo-Gonzalez RR, et al. (November 2009). "PPAR-delta senses and orchestrates clearance of apoptotic cells to promote tolerance". Nature Medicine. 15 (11): 1266–1272. doi:10.1038/nm.2048. PMC 2783696. PMID 19838202.
- ^ Roszer T, Menéndez-Gutiérrez MP, Lefterova MI, Alameda D, Núñez V, Lazar MA, et al. (January 2011). "Autoimmune kidney disease and impaired engulfment of apoptotic cells in mice with macrophage peroxisome proliferator-activated receptor gamma or retinoid X receptor alpha deficiency". Journal of Immunology. 186 (1): 621–631. doi:10.4049/jimmunol.1002230. PMC 4038038. PMID 21135166.
- ^ Singh RP, Waldron RT, Hahn BH (July 2012). "Genes, tolerance and systemic autoimmunity". Autoimmunity Reviews. 11 (9): 664–669. doi:10.1016/j.autrev.2011.11.017. PMC 3306516. PMID 22155015.
- ^ Böhm I (May 2003). "Disruption of the cytoskeleton after apoptosis induction with autoantibodies". Autoimmunity. 36 (3): 183–189. doi:10.1080/0891693031000105617. PMID 12911286. S2CID 37887253.
- ^ Swart JF, Delemarre EM, van Wijk F, Boelens JJ, Kuball J, van Laar JM, Wulffraat NM (April 2017). "Haematopoietic stem cell transplantation for autoimmune diseases". Nature Reviews. Rheumatology. 13 (4): 244–256. doi:10.1038/nrrheum.2017.7. PMID 28228650. S2CID 21264933.
- ^ Drew L (2021-07-14). "How stem cells could fix type 1 diabetes". Nature. 595 (7867): S64–S66. Bibcode:2021Natur.595S..64D. doi:10.1038/d41586-021-01842-x. PMID 34262205. S2CID 235907766.
- ^ Maverakis E, Kim K, Shimoda M, Gershwin ME, Patel F, Wilken R, et al. (February 2015). "Glycans in the immune system and The Altered Glycan Theory of Autoimmunity: a critical review". Journal of Autoimmunity. 57: 1–13. doi:10.1016/j.jaut.2014.12.002. PMC 4340844. PMID 25578468.
- ^ Rook GA (February 2012). "Hygiene hypothesis and autoimmune diseases". Clinical Reviews in Allergy & Immunology. 42 (1): 5–15. doi:10.1007/s12016-011-8285-8. PMID 22090147. S2CID 15302882.
- ^ a b c d e f g Harrison SR, Li D, Jeffery LE, Raza K, Hewison M (January 2020). "Vitamin D, Autoimmune Disease and Rheumatoid Arthritis". Calcified Tissue International. 106 (1): 58–75. doi:10.1007/s00223-019-00577-2. PMC 6960236. PMID 31286174.
- ^ a b Lucas RM, Rodney Harris RM (August 2018). "On the Nature of Evidence and 'Proving' Causality: Smoking and Lung Cancer vs. Sun Exposure, Vitamin D and Multiple Sclerosis". International Journal of Environmental Research and Public Health. 15 (8): 1726. doi:10.3390/ijerph15081726. PMC 6121485. PMID 30103527.
Further reading
[edit]- Vinay K, Abbas AK, Fausto M, Aster J (2010). Robbins and Cotran Pathologic Basis of Disease (8th ed.). Elsevier. p. 1464. ISBN 978-1-4160-3121-5.
- Asherson R (ed.). Handbook of Systemic Autoimmune Diseases. Elsevier.
External links
[edit]- Autoimmune disorders at Curlie
 Media related to Autoimmune diseases and disorders at Wikimedia Commons
Media related to Autoimmune diseases and disorders at Wikimedia Commons
| Type I/allergy/atopy (IgE) |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Type II/ADCC |
| ||||||||
| Type III (Immune complex) |
| ||||||||
| Type IV/cell-mediated (T cells) |
| ||||||||
| Unknown/ multiple |
| ||||||||