| |
| Names | |
|---|---|
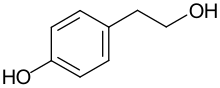
| Preferred IUPAC name
4-(2-Hydroxyethyl)phenol | |
| Other names
p-Hydroxyphenethyl alcohol
2-(4-Hydroxyphenyl)ethanol 4-Hydroxyphenylethanol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.210 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H10O2 | |
| Molar mass | 138.164 g/mol |
| Melting point | 91 to 92 °C (196 to 198 °F; 364 to 365 K) |
| Boiling point | 158 °C (316 °F; 431 K) at 4 Torr |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tyrosol is an organic compound with the formula HOC6H4CH2CH2OH. Classified as a phenylethanoid, i.e. a derivative of phenethyl alcohol, It is found in a variety of natural sources. The compound is colorless solid. The principal source in the human diet is olive oil.[1][2]