GLM
| Distribution | Loglikelihood | Deviance | Valid Links | ||||
|---|---|---|---|---|---|---|---|
| Poisson | Id, ln, sqrt | ||||||
| Gaussian | Id, ln, power | ||||||
| Gamma | |||||||
| Bernoulli | |||||||
| Inverse Gaussian | |||||||
| Negative Binomial |
Shortcuts
| Distribution | Loglikelihood | Deviance | Valid Links | ||||
|---|---|---|---|---|---|---|---|
| Poisson | Id, ln, sqrt | ||||||
| Gaussian | Id, ln, power | ||||||
| Gamma | |||||||
| Bernoulli | |||||||
| Inverse Gaussian | |||||||
| Negative Binomial |
Factor analytic techniques in the most general form seek to model p observed variables as a linear combination of k usually latent or unobserved variables plus orthogonal error variance:
This can be formulated as the modeling of the observed sample covariance, denoted with the covariance implied by the factor analytic model . The model can be specified with a general class of fitting functions, the GLS functions of the general form for and which provided it provided V is a consistent estimator of the asymptotic covariance of the sample covariance(i.e. ), is asymptotically equivalent to estimation by maximum likelihood under normality assumptions, with the fitting function being
A common constraint, usually imposed under the name of Exploratory Factor Analysis(EFA), is The model without this constraint is usually called Confirmatory Factor Analysis(CFA)
Due to orthogonality constraints, EFA solutions are not unique, and is invariant under rotation by an orthogonal matrix. These rotations may be used to obtain a loadings matrices that has desirable properties for the given application(i.e. loadings forced towards zero or one).
Factor analysis is frequently confused with principal component analysis, but it differs as it is a probabilistic rather than a deterministic model, as PCA does not have an underlying generative model.
Fitting of the model by newton-raphson algorithms or MM algorithms(e.g. Expectation Maximization, or EMCM) usually require at least first order derivatives. These are, for a parameter vector
The inverse of the second matrix represents the observed fisher information matrix, , while the expected information can be expressed as
Popular methods of estimation include Lawley and Maxwell's maximum likelihood algorithm,[1] Rubin and Thayer's Expectation Maximization Algorithm[2], or generalized least squares.[3]
The etiology of bipolar disorder is unknown. The overall heritability of bipolar is estimated at 79%-93%, and first degree relatives of bipolar probands have a relative risk of developing bipolar around 7-10. While the heritability is high, no specific genes have been conclusively associated with bipolar, and a number of hypothesis have been posited to explain this fact. The polygenic common rare variant hypothesis suggests that a large number of risk conferring genes are carried in a population, and that a disease manifests when a person has a sufficient number of these genes. The multiple rare variant model suggests that multiple genes that are rare in the population are capable of causing a disease, and that carrying one or a few can lead to disease.
A number of genome wide associations have been reported, including CACNA1C and ODZ4. Less consistently reported loci include ANK3 and NCAN, ITIH1, ITIH3 and NEK4. Significant overlaps with schizophrenia have been reported at CACNA1C, ITIH, ANK3, and ZNF804A. This overlap is congruent with the observation that relatives of probands with schizophrenia are at higher risk for bipolar disorder and vice versa. In light of associations between bipolar and circadian abnormalities(such as decreased need for sleep and increased sleep latency), polymorphisms in the CLOCK gene have been tested for association, although findings have been inconsistent.[6] Other circadian genes associated with bipolar at relaxed significance thresholds include ARTNL, RORB, and DEC1.[7]
Structural neuroimaging studies consistently report increased frequency of white matter hyperintensities in people with bipolar. However, whether or not the lesions play a causative role is unknown. It is possible that they are a result of secondary factors, such as in increased risk of cardiovascular disease in bipolar. On the other hand, the observation of reduced white matter integrity in frontal-subcortical regions makes it possible that these hyperintensities play a role dysfunction between limbic and cortical regions. Global brain volume and morphology are normal in bipolar. Regional deficits in volume have been reported in ventrolateral and dorsolateral prefrontal regions. Based on this, it has been suggested that reduced limbic regulation by prefrontal regions plays a role in bipolar. Findings related to the volume of the basal ganglia have been inconsistent.
Functional neuroimaging studies, contrary to structural studies, consistently find hyperactivation of the basal ganglia, amygdala, and thalamus. Prefrontal abnormalities are less consistently reported, although hyperactivation in the ventral prefrontal cortex is a fairly consistent finding.[8] Mania is generally associated with frontal/ventral hypoactivation, while depression is generally associated with the opposite. A degree of lateralization with regard to abnormalities has been reported, with mania being associated with the right hemisphere, and depression the left. Trait abnormalities in euthymic patients have been observed, including hypoactivity in the ventral prefrontal cortex, and hyperactvity in the amygdala.[9]
Significant overlap in GWAS studies of bipolar and schizophrenia have been observed at ANK3, CACNA1C, ZNF804A, and ITIH3-ITIH4. Studies of brain structure and function in Schizophrenia have included longitudinal, patient-control constrast, and at-risk study designs. Schizophrenia is associated with reduced volumes, usually of the magnitude 5-10%. Robust structural findings include 1)Ventricular enlargement, 2)Reduced whole brain volume, 3)Reduced GMV of the medial temporal lobe extending to the hippocampus and amygdala, 4)Reduced GMV in the STG. thirty-five out of forty-six studies report significant STG volume reduction, and eighteen out of thirty studies reported correlations between STG volume and severity of auditory hallucinations or though disorders. Thirteen studies have reported bilateral thalamic reductions, including studies on both first episodes and chronic patients. Grey matter reductions are particularly evident in the anterior ingulate, medial and inferior frontal lobes, and anterior insula. Bipolar parients and Schizophrenic pateints exhibit significant overlaps in the ACC, mPFC, lPFC, and bilateral insula, only differing in magnitude. The Cerebellum, Corpus Callosum, and Basal Ganglia are frequently observed to be structurally abnormal, although the findings as well as direction of changes is inconsistent. Longitudinal neuroimaging suggests that schizophrenia appears to be more of a neurodevelopment disorder, rather than a neurodegenerative disorder.[12] Schizophrenics exhibit hypoactivity in the dlPFC during executive function tasks, frequently extending to the ACC, thalamus, and inferior and posterior parietal cortices. Hyperactivity during executive function tasks have been observed in midline cortical regions, premotor cortcices and the vlPFC. Reduced activation of the dlPFC during episodic memory tasks has been observed. Reality distortion has been assocaited with increased medial temporal blood flow, and decreased prefrontl boodflow in the other two symptom dimensions. Negtative symptoms in schizophrenia are characterized by more pronounced and widespread GMV losses. MTL generally differentiates affective disorders from less affective psychotic disorders. Longitutinally, Schizophrenia is associated with continual reductions in thalamic and prefrontal volumes and less consistend temporal lobe changes. Hippocampal reductions over time are more contested. In those at high risk for psychosis, inferior frontal and superior temporal reductions are associated with transition to psychosis. Magnetic resonance spectroscopy observe first episode increased in Gln and decreased in Glu, followed by accelerated age related decreased in Gln and Glu relative to controls--->Taken to mean first episode increased in Glu--->Increased in Gln+Decreaese in Glu---->Thereafter reductions in Gln+Glu.[13]
While there is no argument that schizophrenia is associated with cognitive deficits which are in turn associated with abnormalities in the prefrontal cortex and medial temporal lobe, the fundemental cognitive processes that malfunctioning are yet to be determined. Endophenotypic and medication status-independent deficits in WM are observed in schizophrenia, regardless of the modality tested(i.e. verbal versus visual). The temporal characteristics of WM deficits in schizophrenia have led some authors to postulate that deficits are encoding related; in line with this, deficits are constant regardless of delay. However, maintence appears to be dysfunctional as well. While "hypofrontality" is a major principal in schizophenia theories, the relationship between WM task deficit appears to follow a biphasic curve, with increased dlPFC signals occuring earlier in schizophrenic patients, indicating less efficient systems of internal representation. Episodic memory also impaired, and evidence suggests that this is most prominently expressed in relational reasoning caused by hippocampal and prefrontal dysfunction. Hedonic, valenced, processing in schizophrenia appears to largely be intact, as ventral-striatal responses and subjective "liking" is not diminished, although loss-avoidance responsivity is impaired. Despite the aforementioned findings, increased anhedonia(negative symptoms) have also been reportedly associated with reduced striatal responsiveness to rewards. While basal ganglia dysfuncton is present in schizophrenia, simple reinforcement learning is not impaired. However, when reinforcement learning paradigms increase in complexity, impairment becomes apparent. While these impairments are fairly consistent, whether or the deficit is a result of online processing mechanisms(e.g. dlPFC mediated WM), associative processes, or both, is unknown. Monetary incentive delay tasks, assesing desire or "wanting" is frequently associated with reduced ventral striatal signaling in schizophrenia. This has been correlated with various symptoms and symptom dimensions, including apathy and negative symptoms in both medication naive and medicated patients. Reduced prediction errors have also been observed.
Neuropathological studies suggest that volumetric deficits in the temporal lobes, prefrontal lobes, and subiculum of those with Schizophrenia can be better attributed to deficits in neuropil(neuronal extensions such as dendrites) rather than fewer cells. Postmortem gene expression studies have observed reductions in protein associated with myelin formation and regulation, which is congruent with the association between NRGN and DISC1 genetic polymorphisms and schizophrenia.[16] Reductions in GAD67 mRNA levels in the prefrontal cortex and general reduction in parvalbumin mRNA have been observed in schizophrenia, being most significant but not limited to prefrontal regions. The reductions in GABAergic neuronal proteins may be linked to the observed reductions of TrKB and BDNF as the latter proteins are involved in the regulation of the former. The reduction may also be linked to reduced excitation of GABAergic neurons, possible due to hypofunction of NMDA receptors.[17]
| Symptom Dimension | Regional Morphological Differences | Regional Functional Differences |
|---|---|---|
| Negative Symptoms |
|
|
| Psychosis |
|
|
| Disorganization |
|
|
The biology of bipolar disorder is unknown. Various abnormalities in studies of brain structure and function have been demonstrated. Various theories have posited dysfunction in monoaminergic regulation, neural circuitry, and mitochondria function as underlying bipolar disorder.
The etiology of bipolar disorder is unknown. The overall heritability of bipolar is estimated at 79%-93%, and first degree relatives of bipolar probands have a relative risk of developing bipolar around 7-10. While the heritability is high, no specific genes have been conclusively associated with bipolar, and a number of hypothesis have been posited to explain this fact. The polygenic common rare variant hypothesis suggests that a large number of risk conferring genes are carried in a population, and that a disease manifests when a person has a sufficient number of these genes. The multiple rare variant model suggests that multiple genes that are rare in the population are capable of causing a disease, and that carrying one or a few can lead to disease.
A number of genome wide associations have been reported, including CACNA1C and ODZ4. Less consistently reported loci include ANK3 and NCAN, ITIH1, ITIH3 and NEK4. Significant overlaps with schizophrenia have been reported at CACNA1C, ITIH, ANK3, and ZNF804A. This overlap is congruent with the observation that relatives of probands with schizophrenia are at higher risk for bipolar disorder and vice versa. In light of associations between bipolar and circadian abnormalities(such as decreased need for sleep and increased sleep latency), polymorphisms in the CLOCK gene have been tested for association, although findings have been inconsistent.[21] Other circadian genes associated with bipolar at relaxed significance thresholds include ARTNL, RORB, and DEC1.[22] One meta analysis reported a significant association of the short allele of the serotonin transporter, although the study was specific to european populations.[23] Two polymorphisms in the tryptophan hydroxylase 2 gene have been associated with bipolar disorder.[24]
No significant association exists for the BDNF Val66Met allele and bipolar disorder, except in a subgroup of bipolar II cases.[25]
Structural neuroimaging studies consistently report increased frequency of white matter hyperintensities in people with bipolar. However, whether or not the lesions play a causative role is unknown. It is possible that they are a result of secondary factors, such as in increased risk of cardiovascular disease in bipolar. On the other hand, the observation of reduced white matter integrity in frontal-subcortical regions makes it possible that these hyperintensities play a role dysfunction between limbic and cortical regions. Global brain volume and morphology are normal in bipolar. Regional deficits in volume have been reported in ventrolateral and dorsolateral prefrontal regions. Based on this, it has been suggested that reduced limbic regulation by prefrontal regions plays a role in bipolar. Findings related to the volume of the basal ganglia have been inconsistent.
Functional neuroimaging studies, contrary to structural studies, consistently find hyperactivation of the basal ganglia, amygdala, and thalamus. Prefrontal abnormalities are less consistently reported, although hyperactivation in the ventral prefrontal cortex is a fairly consistent finding.[26] Mania is generally associated with frontal/ventral hypoactivation, while depression is generally associated with the opposite. A degree of lateralization with regard to abnormalities has been reported, with mania being associated with the right hemisphere, and depression the left. Trait abnormalities in euthymic patients have been observed, including hypoactivity in the ventral prefrontal cortex, and hyperactvity in the amygdala.[27] Hyperactivity in the amygdala and hypoactivity in the medial and ventral prefrontal cortex during exposure to emotional stimuli has been interpreted as reflecting dysfunction in emotional regulation circuits. Increased effective connectivity between the amygdala and orbitofrontal cortex, and elevated striatal responsiveness during reward tasks have been interpreted as hyper-responsiveness in positive emotion and reward circuitry. The abnormal activity in these circuits has been observed in non-emotional tasks, and is congruent with changes in grey and white matter in these circuits.[28] Neural response during reward tasks differentiates unipolar depression from bipolar depression, with the former being associated with reduced neural response and the latter being associated with elevated neural response.[29]
Meta analyses of structural neuroimaging studies have reported reductions in fronto-insula cortices, the anterior cingulate cortex,[30] ventricular enlargement,[31] increased volumes of the globus pallidus and increased amygdala volume relative to those with schizophrenia,[32] and reduced grey matter in the claustrum and temporal cortex.[33] A significant overlap of bipolar disorder with schizophrenia in grey matter volume reduction occurs in the anterior cingulate cortex, medial prefrontal cortex, lateral prefrontal cortex and bilateral insula.[34]
Regardless of mood state, during response inhibition tasks, people with bipolar disorder underactivate the right inferior frontal gyrus. Changes specific on euthymia include hyperactivations in the left superior temporal gyrus and hypoactivations in the basal ganglia, and changes specific to mania include hyperactivation in the basal ganglia.[35] A meta analysis of fMRI studies reported underactivations in the inferior frontal gyrus and putamen and hyperactivation of the parahippocampus, hippocampus, and amygdala. State specific abnormalities were reported for mania and euthymia. During mania, hypoactivation was significant in the inferior frontal gyrus, while euthymia was associated wit hypoactivation of the lingual gyrus and hyperactivation of the amygdala.[36]
Increased combined glutamine and glutamate(Glx) have been observed globally, regardless of medication status.[37] Increased Glx has been associated with reduced frontal mismatch negativity, interpreted as dysfunction in NMDA signaling.[38] N-acetyl aspartate levels in the basal ganglia are reduced in bipolar disorder, and trends towards increased in the dorsolateral prefrontal cortex. NAA to creatine ratios are reduced in the hippocampus.[39]
Various hypotheses related to monoamines have been proposed. The biogenic amine hypothesis posits general dysregulation of monoamines underlies bipolar and affective disorders. The cholinergic aminergic balance hypothesis posits that an increased ratio of cholingeric activity relative to adrenergic signaling underlies depression, while increased adrenergic signaling relative to cholinergic signaling underlies mania. The permissive hypothesis suggests that serotonin is necessary but not sufficient for affective symptoms, and that reduced serotonergic tone is common to both depression and mania.[40]
Studies of the binding potential of Dopamine receptor D2 and dopamine transporter have been inconsistent but Dopamine receptor D1's binding potential has been observed to be decreased, interpreted as reflecting increased synaptic dopamine in mania. Drugs that release dopamine produce effects similar to mania, further supporting the hypothesis of increase catecholaminergic activity in mania. The binding potential of muscarinic receptors are reduced in vivo during depression, as well as in post mortem studies, supporting the cholinergic aminergic balance hypothesis.[41]
Further evidence for monoamine dysfunction in bipolar comes on studies of neurotransmitter metabolites. Reduced concentration of homovallinic acid, the primary metabolite of dopamine, in the cerebrospinal fluid(CSF) of people with depression is consistently reported. This finding is related to psychomotor retardation and anhedonia. Furthermore, parkinson's disease is associated with high rates of depression, and one case study has reported the abolishment of parkinson's symptoms during manic episodes. The binding potential of VMAT2 is also elevated in bipolar I patients with a history of psychosis, although this finding is inconsistent with finding that valproate increases VMAT2 expression in rodents.[42]
Studies of serotonin's primary metabolite 5-HIAA have been inconsistent,[43] although limited evidence points towards reduced central serotonin signaling in a subgroup of aggressive or suicidal patients.[42] Studies assessing the binding potential of the serotonin transporter or serotonin receptors have also been inconsistent, but generally point towards abnormal serotonin signalling.[44]
Bipolar disorder is associated with elevated basal and dexamethasone elicited cortisol and Adrenocorticotropic hormone(ACTH). These abnormalities are particularly prominent in mania, and are inversely associated with antipsychotic use.[45]
The levels of Gαs but not other g proteins is increased in the frontal, temporal and occipital cortices. The binding of serotonin receptors to g proteins is also elevated globally. Leukocyte and platelet levels of Gαs and Gαi is also elavated in those with bipolar disorder. Downstream targets of g protein signaling is also altered in bipolar disorder. Increased levels of adenylyl cyclase, protein kinase A(PKA), and cyclic adenosine monophosphate induced PKA activity is also elevated. Phosphoinositide signaling is also altered, with elevated levels of phospholipase C, protein kinase C, and Gαq being reported in bipolar.[46]
Glycogen synthase kinase 3 has been implicated in bipolar disorder, as bipolar medications lithium and valproate have been shown to increase ints phosphorylation, thereby inhibiting it. However, postmortem studies hvae not shown any differences in GSK-3 levels or the levels of a downstream target β-catenin.[47]
Some researchers have suggested bipolar disorder is a mitochondrial disease. Some cases of familial chronic progressive external ophthalmoplegia demonstrate increased rates of bipolar disorder before the onset of CPEO, and the higher rate of maternal inheritance patterns support this hypothesis. Further support this hypothesis is the abnormal findings in magnetic resonance spectroscopy studies of brain metabolites in people with bipolar.[48]
A number of abnormalities in GABAergic neurons have been reported in people with bipolar disorder. People with bipolar demonstrate reduced expression of GAD67 in CA3/CA2 subregion of the hippocampus. More extensive reductions of other indicators of GABA function have been reported in in the CA4 and CA1. Abnormal expression of kainate receptors on GABAergic cells have been reported, with reductions in GRIK1 and GRIK2 mRNA in the CA2/CA3 being found in people with bipolar. Decreased levels of HCN channels have also been reported, which, along with abnormal glutamate signaling, could contribute to reduced GABAergic tone in the hippocampus.[49]
The observation of increased Glx in the prefrontal cortex is congruent with the observation of reduced glial cell counts and prefrontal cortex volume, as glia play an important role in glutamate homeostasis.[50]
Elevated levels of IL-6, C-reactive protein(CRP) and TNFα have been reported in bipolar. Levels of some(IL-6 and CRP) but not all (TNFα) may be reduced by treatment. Increases in IL-6 have been reported in mood episodes, regardless of polarity.[51]
| Link | Decreases(-) | Increases(+) | Measure |
|---|---|---|---|
| Kühn 2013
Resting-state brain activity in schizophrenia and major depression: a quantitative meta-analysis. |
|
|
Resting activity |
| Miller 2015 |
Functional activity | ||
| Wang 2016
Serotonin-1A receptor alterations in depression: a meta-analysis of molecular imaging studies |
|
5-HT1A receptor BP | |
| Du 2012
Voxelwise meta-analysis of gray matter reduction in major depressive disorder |
|
Grey matter volume | |
| Hamilton 2012 |
|
|
Functional activity |
| Zhang 2016 |
|
Grey matter volume | |
| Bora 2012 |
|
Grey matter | |
| Lai 2013
Gray matter volume in major depressive disorder: a meta-analysis of voxel-based morphometry studies |
|
Grey matter | |
| Sacher 2012 |
|
|
Decreases(GMV), increases(Glucose metebolism) |
| Gryglewski 2014
Meta-analysis of molecular imaging of serotonin transporters in major depression |
|
5-HTT Binding potential |
| Title | Year | Disorder | Papers(n) | Subjects | Pubmed Link | Full Text Link | Full Text Link | DOI | Increased Activity, GM, WM or BP | Reduced Activity, GM, WM OR BP | Conclusion | Average Subjects |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Functional Decoding and Meta-analytic Connectivity Modeling in Adult Attention-Deficit/Hyperactivity Disorder. | 2016 | ADHD | 24 | - | https://www.ncbi.nlm.nih.gov/pubmed/27569542 | sci-hub.tw/10.1016/j.biopsych.2016.06.014. | sci-hub.la/10.1016/j.biopsych.2016.06.014. | 10.1016/j.biopsych.2016.06.014. | L PUT, L IFG, R CAUD | - | ||
| A systematic review and meta-analysis of tract-based spatial statistics studies regarding attention-deficit/hyperactivity disorder | 2016 | ADHD | 10 | 947 | https://www.ncbi.nlm.nih.gov/pubmed/27450582 | sci-hub.tw/10.1016/j.neubiorev.2016.07.022 | sci-hub.la/10.1016/j.neubiorev.2016.07.022 | 10.1016/j.neubiorev.2016.07.022 | - | SS, Sagital Stratum | 94.7 | |
| Striatal dopamine transporter alterations in ADHD: pathophysiology or adaptation to psychostimulants? A meta-analysis. | 2012 | ADHD | 9 | 169 | https://www.ncbi.nlm.nih.gov/pubmed/22294258 | sci-hub.tw/10.1176/appi.ajp.2011.11060940. | sci-hub.la/10.1176/appi.ajp.2011.11060940. | 10.1176/appi.ajp.2011.11060940. | Striatal DAT | - | 18.8 | |
| Toward systems neuroscience of ADHD: a meta-analysis of 55 fMRI studies. | 2012 | ADHD | 55 | 1542 | https://www.ncbi.nlm.nih.gov/pubmed/22983386 | sci-hub.tw/10.1176/appi.ajp.2012.11101521. | sci-hub.la/10.1176/appi.ajp.2012.11101521. | 10.1176/appi.ajp.2012.11101521. | MCC, Angular | R vlPFC, L INS, L PUT | 28.0 | |
| Diffusion tensor imaging in attention deficit/hyperactivity disorder: a systematic review and meta-analysis | 2012 | ADHD | 9 | 342 | https://www.ncbi.nlm.nih.gov/pubmed/22305957 | sci-hub.tw/10.1016/j.neubiorev.2012.01.003 | sci-hub.la/10.1016/j.neubiorev.2012.01.003 | 10.1016/j.neubiorev.2012.01.003 | 38.0 | |||
| Meta-analysis of structural MRI studies in children and adults with attention deficit hyperactivitydisorder indicates treatment effects | 2012 | ADHD | 11 | 608 | https://www.ncbi.nlm.nih.gov/pubmed/22118249 | sci-hub.tw/10.1111/j.1600-0447.2011.01786.x | sci-hub.la/10.1111/j.1600-0447.2011.01786.x | 10.1111/j.1600-0447.2011.01786.x | 55.3 | |||
| Gray matter volume abnormalities in ADHD: voxel-based meta-analysis exploring the effects of age and stimulant medication | 2011 | ADHD | 14 | 622 | https://www.ncbi.nlm.nih.gov/pubmed/21865529 | sci-hub.tw/10.1176/appi.ajp.2011.11020281 | sci-hub.la/10.1176/appi.ajp.2011.11020281 | 10.1176/appi.ajp.2011.11020281 | 44.4 | |||
| Structural Neuroimaging of Anorexia Nervosa: Future Directions in the Quest for Mechanisms Underlying Dynamic Alterations. | 2018 | AN | - | - | https://www.ncbi.nlm.nih.gov/pubmed/28967386 | sci-hub.tw/10.1016/j.biopsych.2017.08.011 | sci-hub.la/10.1016/j.biopsych.2017.08.011 | 10.1016/j.biopsych.2017.08.011 | - | |||
| Behind binge eating: A review of food-specific adaptations of neurocognitive and neuroimaging tasks. | 2017 | AN | - | - | https://www.ncbi.nlm.nih.gov/pubmed/28363840 | sci-hub.tw/10.1016/j.physbeh.2017.03.037 | sci-hub.la/10.1016/j.physbeh.2017.03.037 | 10.1016/j.physbeh.2017.03.037 | - | |||
| Functional brain alterations in anorexia nervosa: a scoping review. | 2016 | AN | 49 | - | https://www.ncbi.nlm.nih.gov/pubmed/27933159 | sci-hub.tw/10.1186/s40337-016-0118-y | sci-hub.la/10.1186/s40337-016-0118-y | 10.1186/s40337-016-0118-y | - | |||
| A systematic review of resting-state functional-MRI studies in anorexia nervosa: Evidence for functional connectivity impairment in cognitive control and visuospatial and body-signal integration. | 2016 | AN | 15 | 294 | https://www.ncbi.nlm.nih.gov/pubmed/27725172 | sci-hub.tw/10.1016/j.neubiorev.2016.09.032 | sci-hub.la/10.1016/j.neubiorev.2016.09.032 | 10.1016/j.neubiorev.2016.09.032 | 19.6 | |||
| Brain morphological changes in adolescent and adult patients with anorexia nervosa. | 2016 | AN | 29 | 273 | https://www.ncbi.nlm.nih.gov/pubmed/27188331 | sci-hub.tw/10.1007/s00702-016-1567-9 | sci-hub.la/10.1007/s00702-016-1567-9 | 10.1007/s00702-016-1567-9 | 9.4 | |||
| Morphological changes in the brain of acutely ill and weight-recovered patients with anorexia nervosa. A meta-analysis and qualitative review. | 2014 | AN | - | 177 | https://www.ncbi.nlm.nih.gov/pubmed/24365959 | sci-hub.tw/10.1024/1422-4917/a000265 | sci-hub.la/10.1024/1422-4917/a000265 | 10.1024/1422-4917/a000265 | - | |||
| Anorexia nervosa is linked to reduced brain structure in reward and somatosensory regions: a meta-analysis of VBM studies. | 2013 | AN | 9 | 228 | https://www.ncbi.nlm.nih.gov/pubmed/23570420 | sci-hub.tw/10.1186/1471-244X-13-110 | sci-hub.la/10.1186/1471-244X-13-110 | 10.1186/1471-244X-13-110 | 25.3 | |||
| Neural basis of a multidimensional model of body image distortion in anorexia nervosa. | 2012 | AN | - | - | https://www.ncbi.nlm.nih.gov/pubmed/22613629 | sci-hub.tw/10.1016/j.neubiorev.2012.05.003 | sci-hub.la/10.1016/j.neubiorev.2012.05.003 | 10.1016/j.neubiorev.2012.05.003 | - | |||
| Processing of food, body and emotional stimuli in anorexia nervosa: a systematic review and meta-analysis of functional magnetic resonance imaging studies. | 2012 | AN | 15 | - | https://www.ncbi.nlm.nih.gov/pubmed/22945872 | sci-hub.tw/10.1002/erv.2197 | sci-hub.la/10.1002/erv.2197 | 10.1002/erv.2197 | - | |||
| The common traits of the ACC and PFC in anxiety disorders in the DSM-5: meta-analysis of voxel-based morphometry studies. | 2014 | ANX | 24 | 1264 | https://www.ncbi.nlm.nih.gov/pubmed/24676455 | sci-hub.tw/10.1371/journal.pone.0093432 | sci-hub.la/10.1371/journal.pone.0093432 | 10.1371/journal.pone.0093432 | 52.7 | |||
| Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. | 2007 | ANX | 19 | 1674 | https://www.ncbi.nlm.nih.gov/pubmed/17898336 | sci-hub.tw/10.1176/appi.ajp.2007.07030504 | sci-hub.la/10.1176/appi.ajp.2007.07030504 | 10.1176/appi.ajp.2007.07030504 | 88.1 | |||
| Prefrontal Structural and Functional Brain Imaging findings in Antisocial, Violent, and Psychopathic Individuals: A Meta-Analysis | 2009 | ASPD | 43 | 1262 | https://www.ncbi.nlm.nih.gov/pubmed/19833485 | sci-hub.tw/10.1016/j.pscychresns.2009.03.012 | sci-hub.la/10.1016/j.pscychresns.2009.03.012 | 10.1016/j.pscychresns.2009.03.012 | 29.3 | |||
| Neuroimaging in bulimia nervosa and binge eating disorder: a systematic review. | 2018 | BN | 32 | - | https://www.ncbi.nlm.nih.gov/pubmed/29468065 | sci-hub.tw/10.1186/s40337-018-0187-1 | sci-hub.la/10.1186/s40337-018-0187-1 | 10.1186/s40337-018-0187-1 | - | |||
| Inhibitory control in obesity and binge eating disorder: A systematic review and meta-analysis of neurocognitive and neuroimaging studies. | 2016 | BN | 8 | 150 | https://www.ncbi.nlm.nih.gov/pubmed/27381956 | sci-hub.tw/10.1016/j.neubiorev.2016.06.041 | sci-hub.la/10.1016/j.neubiorev.2016.06.041 | 10.1016/j.neubiorev.2016.06.041 | 18.8 | |||
| Common and distinct patterns of grey-matter volume alteration in major depression and bipolar disorder: evidence from voxel-based meta-analysis. | 2017 | BP | 86 | 6058 | https://www.ncbi.nlm.nih.gov/pubmed/27217146 | sci-hub.tw/10.1038/mp.2016.72 | sci-hub.la/10.1038/mp.2016.72 | 10.1038/mp.2016.72 | S dmPFC | pgACC, sgACC, INS | 70.4 | |
| Meta-analysis of functional magnetic resonance imaging studies of timing and cognitive control in schizophrenia and bipolar disorder: Evidence of a primary time deficit. | 2017 | BP | 22 | 772 | https://www.ncbi.nlm.nih.gov/pubmed/28169089 | sci-hub.tw/10.1016/j.schres.2017.01.039 | sci-hub.la/10.1016/j.schres.2017.01.039 | 10.1016/j.schres.2017.01.039 | 35.1 | |||
| Brain functional effects of psychopharmacological treatments in bipolar disorder. | 2016 | BP | 140 | - | https://www.ncbi.nlm.nih.gov/pubmed/27617780 | sci-hub.tw/10.1016/j.euroneuro.2016.06.006 | sci-hub.la/10.1016/j.euroneuro.2016.06.006 | 10.1016/j.euroneuro.2016.06.006 | - | |||
| Subcortical volumetric abnormalities in bipolar disorder. | 2016 | BP | 1 | 4304 | https://www.ncbi.nlm.nih.gov/pubmed/26857596 | sci-hub.tw/10.1038/mp.2015.227 | sci-hub.la/10.1038/mp.2015.227 | 10.1038/mp.2015.227 | 4304.0 | |||
| Voxel-Based Meta-Analytical Evidence of Structural Disconnectivity in Major Depression and Bipolar Disorder. | 2016 | BP | 40 | 2429 | https://www.ncbi.nlm.nih.gov/pubmed/25891219 | sci-hub.tw/10.1016/j.biopsych.2015.03.004 | sci-hub.la/10.1016/j.biopsych.2015.03.004 | 10.1016/j.biopsych.2015.03.004 | 60.7 | |||
| A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. | 2014 | BP | - | - | https://www.ncbi.nlm.nih.gov/pubmed/24626773 | sci-hub.tw/10.1176/appi.ajp.2014.13081008 | sci-hub.la/10.1176/appi.ajp.2014.13081008 | 10.1176/appi.ajp.2014.13081008 | - | |||
| Elevated reward-related neural activation as a unique biological marker of bipolar disorder: assessment and treatment implications. | 2014 | BP | - | - | https://www.ncbi.nlm.nih.gov/pubmed/25241675 | sci-hub.tw/10.1016/j.brat.2014.08.011 | sci-hub.la/10.1016/j.brat.2014.08.011 | 10.1016/j.brat.2014.08.011 | - | |||
| Developmental meta-analyses of the functional neural correlates of bipolar disorder. | 2014 | BP | 94 | 3896 | https://www.ncbi.nlm.nih.gov/pubmed/25100166 | sci-hub.tw/10.1001/jamapsychiatry.2014.660 | sci-hub.la/10.1001/jamapsychiatry.2014.660 | 10.1001/jamapsychiatry.2014.660 | 41.4 | |||
| Could glutamate spectroscopy differentiate bipolar depression from unipolar? | 2014 | BP | 11 | 431 | https://www.ncbi.nlm.nih.gov/pubmed/25082118 | sci-hub.tw/10.1016/j.jad.2014.05.019 | sci-hub.la/10.1016/j.jad.2014.05.019 | 10.1016/j.jad.2014.05.019 | 39.2 | |||
| Gray matter abnormalities as brain structural vulnerability factors for bipolar disorder: A review of neuroimaging studies of individuals at high genetic risk for bipolar disorder. | 2013 | BP | - | - | https://www.ncbi.nlm.nih.gov/pubmed/23864160 | sci-hub.tw/10.1177/0004867413496482 | sci-hub.la/10.1177/0004867413496482 | 10.1177/0004867413496482 | - | |||
| Functional neuroanatomy of response inhibition in bipolar disorders--combined voxel based and cognitive performance meta-analysis. | 2013 | BP | 30 | 1302 | https://www.ncbi.nlm.nih.gov/pubmed/24070910 | sci-hub.tw/10.1016/j.jpsychires.2013.08.015 | sci-hub.la/10.1016/j.jpsychires.2013.08.015 | 10.1016/j.jpsychires.2013.08.015 | 43.4 | |||
| Evidence of diagnostic specificity in the neural correlates of facial affect processing in bipolar disorder and schizophrenia: a meta-analysis of functional imaging studies. | 2013 | BP | 29 | 750 | https://www.ncbi.nlm.nih.gov/pubmed/22874625 | sci-hub.tw/10.1017/S0033291712001432 | sci-hub.la/10.1017/S0033291712001432 | 10.1017/S0033291712001432 | 25.9 | |||
| A systematic review and meta-analysis of proton magnetic resonance spectroscopy and mismatch negativity in bipolar disorder. | 2013 | BP | 15 | 368 | https://www.ncbi.nlm.nih.gov/pubmed/23968965 | sci-hub.tw/10.1016/j.euroneuro.2013.07.007 | sci-hub.la/10.1016/j.euroneuro.2013.07.007 | 10.1016/j.euroneuro.2013.07.007 | 24.5 | |||
| Mapping vulnerability to bipolar disorder: a systematic review and meta-analysis of neuroimaging studies. | 2012 | BP | 37 | 2254 | https://www.ncbi.nlm.nih.gov/pubmed/22297067 | sci-hub.tw/10.1503/jpn.110061 | sci-hub.la/10.1503/jpn.110061 | 10.1503/jpn.110061 | 60.9 | |||
| Neurometabolites in schizophrenia and bipolar disorder - a systematic review and meta-analysis. | 2012 | BP | 43 | 1461 | https://www.ncbi.nlm.nih.gov/pubmed/22981426 | sci-hub.tw/10.1016/j.pscychresns.2012.02.003 | sci-hub.la/10.1016/j.pscychresns.2012.02.003 | 10.1016/j.pscychresns.2012.02.003 | 34.0 | |||
| Common and distinct neural correlates of emotional processing in Bipolar Disorder and Major Depressive Disorder: a voxel-based meta-analysis of functional magnetic resonance imaging studies. | 2012 | BP | 20 | 701 | https://www.ncbi.nlm.nih.gov/pubmed/21820878 | sci-hub.tw/10.1016/j.euroneuro.2011.07.003 | sci-hub.la/10.1016/j.euroneuro.2011.07.003 | 10.1016/j.euroneuro.2011.07.003 | 35.1 | |||
| Grey matter differences in bipolar disorder: a meta-analysis of voxel-based morphometry studies. | 2012 | BP | 8 | 520 | https://www.ncbi.nlm.nih.gov/pubmed/22420589 | sci-hub.tw/10.1111/j.1399-5618.2012.01000.x | sci-hub.la/10.1111/j.1399-5618.2012.01000.x | 10.1111/j.1399-5618.2012.01000.x | 65.0 | |||
| A quantitative meta-analysis of fMRI studies in bipolar disorder. | 2011 | BP | 65 | 2114 | https://www.ncbi.nlm.nih.gov/pubmed/21320248 | sci-hub.tw/10.1111/j.1399-5618.2011.00893.x | sci-hub.la/10.1111/j.1399-5618.2011.00893.x | 10.1111/j.1399-5618.2011.00893.x | 32.5 | |||
| Toward a functional neuroanatomical signature of bipolar disorder: quantitative evidence from the neuroimaging literature. | 2011 | BP | 55 | 1584 | https://www.ncbi.nlm.nih.gov/pubmed/21676596 | sci-hub.tw/10.1016/j.pscychresns.2011.02.011 | sci-hub.la/10.1016/j.pscychresns.2011.02.011 | 10.1016/j.pscychresns.2011.02.011 | 28.8 | |||
| Neuroimaging-based markers of bipolar disorder: evidence from two meta-analyses. | 2011 | BP | 28 | 1561 | https://www.ncbi.nlm.nih.gov/pubmed/21470688 | sci-hub.tw/10.1016/j.jad.2011.03.016 | sci-hub.la/10.1016/j.jad.2011.03.016 | 10.1016/j.jad.2011.03.016 | 55.8 | |||
| Voxelwise meta-analysis of gray matter abnormalities in bipolar disorder. | 2010 | BP | 21 | 1430 | https://www.ncbi.nlm.nih.gov/pubmed/20303066 | sci-hub.tw/10.1016/j.biopsych.2010.01.020 | sci-hub.la/10.1016/j.biopsych.2010.01.020 | 10.1016/j.biopsych.2010.01.020 | 68.1 | |||
| Correlation between amygdala volume and age in bipolar disorder - a systematic review and meta-analysis of structural MRI studies. | 2010 | BP | 13 | 877 | https://www.ncbi.nlm.nih.gov/pubmed/20226638 | sci-hub.tw/10.1016/j.pscychresns.2009.09.004 | sci-hub.la/10.1016/j.pscychresns.2009.09.004 | 10.1016/j.pscychresns.2009.09.004 | 67.5 | |||
| Magnetic resonance imaging studies in bipolar disorder and schizophrenia: meta-analysis. | 2009 | BP | 65 | 1384 | https://www.ncbi.nlm.nih.gov/pubmed/19721106 | sci-hub.tw/10.1192/bjp.bp.108.059717 | sci-hub.la/10.1192/bjp.bp.108.059717 | 10.1192/bjp.bp.108.059717 | 21.3 | |||
| Meta-analysis, database, and meta-regression of 98 structural imaging studies in bipolar disorder. | 2008 | BP | 98 | 8196 | https://www.ncbi.nlm.nih.gov/pubmed/18762588 | sci-hub.tw/10.1001/archpsyc.65.9.1017 | sci-hub.la/10.1001/archpsyc.65.9.1017 | 10.1001/archpsyc.65.9.1017 | 83.6 | |||
| Meta-analysis of amygdala volumes in children and adolescents with bipolar disorder. | 2008 | BP | 11 | 553 | https://www.ncbi.nlm.nih.gov/pubmed/18827720 | sci-hub.tw/10.1097/CHI.0b013e318185d299 | sci-hub.la/10.1097/CHI.0b013e318185d299 | 10.1097/CHI.0b013e318185d299 | 50.3 | |||
| Neurochemical alterations of the brain in bipolar disorder and their implications for pathophysiology: a systematic review of the in vivo proton magnetic resonance spectroscopy findings. | 2006 | BP | - | - | https://www.ncbi.nlm.nih.gov/pubmed/16677749 | sci-hub.tw/10.1016/j.pnpbp.2006.03.012 | sci-hub.la/10.1016/j.pnpbp.2006.03.012 | 10.1016/j.pnpbp.2006.03.012 | - | |||
| Neural Correlates of Disturbed Emotion Processing in Borderline Personality Disorder: A Multimodal Meta-Analysis. | 2016 | BPD | 29 | 1115 | https://www.ncbi.nlm.nih.gov/pubmed/25935068 | sci-hub.tw/10.1016/j.biopsych.2015.03.027 | sci-hub.la/10.1016/j.biopsych.2015.03.027 | 10.1016/j.biopsych.2015.03.027 | 38.4 | |||
| Mapping the brain correlates of borderline personality disorder: A functional neuroimaging meta-analysis of resting state studies. | 2016 | BPD | 7 | 299 | https://www.ncbi.nlm.nih.gov/pubmed/27552444 | sci-hub.tw/10.1016/j.jad.2016.07.025 | sci-hub.la/10.1016/j.jad.2016.07.025 | 10.1016/j.jad.2016.07.025 | 42.7 | |||
| Meta-analysis of molecular imaging of serotonin transporters in ecstasy/polydrug users. | 2016 | DA | 7 | 305 | https://www.ncbi.nlm.nih.gov/pubmed/26855234 | sci-hub.tw/10.1016/j.neubiorev.2016.02.003 | sci-hub.la/10.1016/j.neubiorev.2016.02.003 | 10.1016/j.neubiorev.2016.02.003 | 43.6 | |||
| A Systematic Review and Meta-analysis of Neuroimaging in Oppositional Defiant Disorder (ODD) and Conduct Disorder (CD) Taking Attention-Deficit Hyperactivity Disorder (ADHD) Into Account. | 2016 | DBD | 29 | 838 | https://www.ncbi.nlm.nih.gov/pubmed/26846227 | sci-hub.tw/10.1007/s11065-015-9315-8 | sci-hub.la/10.1007/s11065-015-9315-8 | 10.1007/s11065-015-9315-8 | 28.9 | |||
| Meta-Analysis of fMRI Studies of Disruptive Behavior Disorders. | 2016 | DBD | 24 | 636 | https://www.ncbi.nlm.nih.gov/pubmed/27523497 | sci-hub.tw/10.1176/appi.ajp.2016.15081089 | sci-hub.la/10.1176/appi.ajp.2016.15081089 | 10.1176/appi.ajp.2016.15081089 | 26.5 | |||
| Alterations in emotion generation and regulation neurocircuitry in depression and eating disorders: A comparative review of structural and functional neuroimaging studies. | 2016 | ED | - | - | https://www.ncbi.nlm.nih.gov/pubmed/27422451 | sci-hub.tw/10.1016/j.neubiorev.2016.07.011 | sci-hub.la/10.1016/j.neubiorev.2016.07.011 | 10.1016/j.neubiorev.2016.07.011 | - | |||
| A systematic review of temporal discounting in eating disorders and obesity: Behavioural and neuroimaging findings. | 2016 | ED | 31 | 4546 | https://www.ncbi.nlm.nih.gov/pubmed/27693228 | sci-hub.tw/10.1016/j.neubiorev.2016.09.024 | sci-hub.la/10.1016/j.neubiorev.2016.09.024 | 10.1016/j.neubiorev.2016.09.024 | 146.6 | |||
| Neuroimaging and neuromodulation approaches to study eating behavior and prevent and treat eating disorders and obesity. | 2015 | ED | - | - | https://www.ncbi.nlm.nih.gov/pubmed/26110109 | sci-hub.tw/10.1016/j.nicl.2015.03.016 | sci-hub.la/10.1016/j.nicl.2015.03.016 | 10.1016/j.nicl.2015.03.016 | - | |||
| Positron emission tomography studies in eating disorders: multireceptor brain imaging, correlates with behavior and implications for pharmacotherapy. | 2005 | ED | - | - | https://www.ncbi.nlm.nih.gov/pubmed/16243652 | sci-hub.tw/10.1016/j.nucmedbio.2005.06.011 | sci-hub.la/10.1016/j.nucmedbio.2005.06.011 | 10.1016/j.nucmedbio.2005.06.011 | - | |||
| Melancholy, anhedonia, apathy: the search for separable behaviors and neural circuits in depression. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/29529482 | sci-hub.tw/10.1016/j.conb.2018.02.018 | sci-hub.la/10.1016/j.conb.2018.02.018 | 10.1016/j.conb.2018.02.018 | - | |||
| Circuit-based frameworks of depressive behaviors: The role of reward circuitry and beyond. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/29309799 | sci-hub.tw/10.1016/j.pbb.2017.12.010 | sci-hub.la/10.1016/j.pbb.2017.12.010 | 10.1016/j.pbb.2017.12.010 | - | |||
| Treatment resistant depression: A multi-scale, systems biology approach. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/28859997 | sci-hub.tw/10.1016/j.neubiorev.2017.08.019 | sci-hub.la/10.1016/j.neubiorev.2017.08.019 | 10.1016/j.neubiorev.2017.08.019 | - | |||
| Dopamine System Dysregulation in Major Depressive Disorders. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/29106542 | sci-hub.tw/10.1093/ijnp/pyx056 | sci-hub.la/10.1093/ijnp/pyx056 | 10.1093/ijnp/pyx056 | - | |||
| Progress in understanding mood disorders: optogenetic dissection of neural circuits. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/23682971 | sci-hub.tw/10.1111/gbb.12049 | sci-hub.la/10.1111/gbb.12049 | 10.1111/gbb.12049 | - | |||
| Presentation and Neurobiology of Anhedonia in Mood Disorders: Commonalities and Distinctions. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/29520717 | sci-hub.tw/10.1007/s11920-018-0877-z | sci-hub.la/10.1007/s11920-018-0877-z | 10.1007/s11920-018-0877-z | - | |||
| Lateral habenula in the pathophysiology of depression. | 2018 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/29175713 | sci-hub.tw/10.1016/j.conb.2017.10.024 | sci-hub.la/10.1016/j.conb.2017.10.024 | 10.1016/j.conb.2017.10.024 | - | |||
| Neuroimaging genomic studies in major depressive disorder: A systematic review. | 2018 | MDD | 64 | - | https://www.ncbi.nlm.nih.gov/pubmed/29476595 | sci-hub.tw/10.1111/cns.12829 | sci-hub.la/10.1111/cns.12829 | 10.1111/cns.12829 | - | |||
| Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. | 2017 | MDD | 20 | 10105 | https://www.ncbi.nlm.nih.gov/pubmed/27137745 | sci-hub.tw/10.1038/mp.2016.60 | sci-hub.la/10.1038/mp.2016.60 | 10.1038/mp.2016.60 | 505.3 | |||
| Subcortical brain structure and suicidal behaviour in major depressive disorder: a meta-analysis from the ENIGMA-MDD working group. | 2017 | MDD | 1 | 3097 | https://www.ncbi.nlm.nih.gov/pubmed/28463239 | sci-hub.tw/10.1038/tp.2017.84 | sci-hub.la/10.1038/tp.2017.84 | 10.1038/tp.2017.84 | 3097.0 | |||
| Disorganization of white matter architecture in major depressive disorder: a meta-analysis of diffusion tensor imaging with tract-based spatial statistics | 2017 | MDD | 18 | 1122 | https://www.ncbi.nlm.nih.gov/pubmed/26906716 | sci-hub.tw/10.1038/srep21825 | sci-hub.la/10.1038/srep21825 | 10.1038/srep21825 | 62.3 | |||
| Computational meta-analysis of statistical parametric maps in major depression | 2017 | MDD | 12 | 1052 | https://www.ncbi.nlm.nih.gov/pubmed/26854015 | sci-hub.tw/10.1002/hbm.23108 | sci-hub.la/10.1002/hbm.23108 | 10.1002/hbm.23108 | 87.7 | |||
| Altered Brain Activity in Unipolar Depression Revisited: Meta-analyses of Neuroimaging Studies. | 2017 | MDD | 57 | 2116 | https://www.ncbi.nlm.nih.gov/pubmed/27829086 | sci-hub.tw/10.1001/jamapsychiatry.2016.2783 | sci-hub.la/10.1001/jamapsychiatry.2016.2783 | 10.1001/jamapsychiatry.2016.2783 | 37.1 | |||
| Characterization of brain blood flow and the amplitude of low-frequency fluctuations in major depressive disorder: A multimodal meta-analysis. | 2017 | MDD | 16 | 972 | https://www.ncbi.nlm.nih.gov/pubmed/28068619 | sci-hub.tw/10.1016/j.jad.2016.12.032 | sci-hub.la/10.1016/j.jad.2016.12.032 | 10.1016/j.jad.2016.12.032 | 60.8 | |||
| Intrinsic cerebral activity at resting state in adults with major depressive disorder: A meta-analysis. | 2017 | MDD | 15 | 859 | https://www.ncbi.nlm.nih.gov/pubmed/28174129 | sci-hub.tw/10.1016/j.pnpbp.2017.02.001 | sci-hub.la/10.1016/j.pnpbp.2017.02.001 | 10.1016/j.pnpbp.2017.02.001 | 57.3 | |||
| Microstructural brain abnormalities in medication-free patients with major depressive disorder: a systematic review and meta-analysis of diffusion tensor imaging | 2017 | MDD | 15 | 864 | https://www.ncbi.nlm.nih.gov/pubmed/27780031 | sci-hub.tw/10.1503/jpn.150341 | sci-hub.la/10.1503/jpn.150341 | 10.1503/jpn.150341 | 57.6 | |||
| Corticostriatal circuitry in regulating diseases characterized by intrusive thinking. | 2016 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/27069381 | https://www.ncbi.nlm.nih.gov/pubmed/27069381 | https://www.ncbi.nlm.nih.gov/pubmed/27069381 | https://www.ncbi.nlm.nih.gov/pubmed/27069381 | - | |||
| Reward processing by the lateral habenula in normal and depressive behaviors. | 2016 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/25157511 | sci-hub.tw/10.1038/nn.3779 | sci-hub.la/10.1038/nn.3779 | 10.1038/nn.3779 | - | |||
| Subcortical brain alterations in major depressive disorder: findings from the ENIGMA Major Depressive Disorder working group. | 2016 | MDD | 1 | 8927 | https://www.ncbi.nlm.nih.gov/pubmed/26122586 | sci-hub.tw/10.1038/mp.2015.69 | sci-hub.la/10.1038/mp.2015.69 | 10.1038/mp.2015.69 | 8927.0 | |||
| Shared white matter alterations across emotional disorders: A voxel-based meta-analysis of fractional anisotropy. | 2016 | MDD | 37 | 1954 | https://www.ncbi.nlm.nih.gov/pubmed/27995068 | sci-hub.tw/10.1016/j.nicl.2016.09.001 | sci-hub.la/10.1016/j.nicl.2016.09.001 | 10.1016/j.nicl.2016.09.001 | 52.8 | |||
| Functional alterations of fronto-limbic circuit and default mode network systems in first-episode, drug-naïve patients with major depressive disorder: A meta-analysis of resting-state fMRI data. | 2016 | MDD | 31 | 908 | https://www.ncbi.nlm.nih.gov/pubmed/27639862 | sci-hub.tw/10.1016/j.jad.2016.09.005 | sci-hub.la/10.1016/j.jad.2016.09.005 | 10.1016/j.jad.2016.09.005 | 29.3 | |||
| Essential brain structural alterations in major depressive disorder: A voxel-wise meta-analysis on first episode, medication-naive patients. | 2016 | MDD | 10 | 669 | https://www.ncbi.nlm.nih.gov/pubmed/27100056 | sci-hub.tw/10.1016/j.jad.2016.04.001 | sci-hub.la/10.1016/j.jad.2016.04.001 | 10.1016/j.jad.2016.04.001 | 66.9 | |||
| Serotonin-1A receptor alterations in depression: a meta-analysis of molecular imaging studies. | 2016 | MDD | 10 | 479 | https://www.ncbi.nlm.nih.gov/pubmed/27623971 | sci-hub.tw/10.1186/s12888-016-1025-0 | sci-hub.la/10.1186/s12888-016-1025-0 | 10.1186/s12888-016-1025-0 | 47.9 | |||
| Abnormal reward functioning across substance use disorders and major depressive disorder: Considering reward as a transdiagnostic mechanism. | 2015 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/25655926 | sci-hub.tw/10.1016/j.ijpsycho.2015.01.011 | sci-hub.la/10.1016/j.ijpsycho.2015.01.011 | 10.1016/j.ijpsycho.2015.01.011 | - | |||
| Reinforcement learning in depression: A review of computational research. | 2015 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/25979140 | sci-hub.tw/10.1016/j.neubiorev.2015.05.005 | sci-hub.la/10.1016/j.neubiorev.2015.05.005 | 10.1016/j.neubiorev.2015.05.005 | - | |||
| Role of the Brain's Reward Circuitry in Depression: Transcriptional Mechanisms. | 2015 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/26472529 | sci-hub.tw/10.1016/bs.irn.2015.07.003 | sci-hub.la/10.1016/bs.irn.2015.07.003 | 10.1016/bs.irn.2015.07.003 | - | |||
| The serotonin transporter in depression: Meta-analysis of in vivo and post mortem findings and implications for understanding and treating depression. | 2015 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/26281039 | sci-hub.tw/10.1016/j.jad.2015.07.034 | sci-hub.la/10.1016/j.jad.2015.07.034 | 10.1016/j.jad.2015.07.034 | - | |||
| The Brain's Response to Reward Anticipation and Depression in Adolescence: Dimensionality, Specificity, and Longitudinal Predictions in a Community-Based Sample. | 2015 | MDD | 1 | 1576 | https://www.ncbi.nlm.nih.gov/pubmed/26085042 | sci-hub.tw/10.1176/appi.ajp.2015.14101298 | sci-hub.la/10.1176/appi.ajp.2015.14101298 | 10.1176/appi.ajp.2015.14101298 | 1576.0 | |||
| Large-Scale Network Dysfunction in Major Depressive Disorder: A Meta-analysis of Resting-State Functional Connectivity. | 2015 | MDD | 27 | 1074 | https://www.ncbi.nlm.nih.gov/pubmed/25785575 | sci-hub.tw/10.1001/jamapsychiatry.2015.0071 | sci-hub.la/10.1001/jamapsychiatry.2015.0071 | 10.1001/jamapsychiatry.2015.0071 | 39.8 | |||
| Voxel-wise meta-analyses of brain blood flow and local synchrony abnormalities in medication-free patients with major depressive disorder. | 2015 | MDD | 23 | 1068 | https://www.ncbi.nlm.nih.gov/pubmed/25853283 | sci-hub.tw/10.1503/jpn.140119 | sci-hub.la/10.1503/jpn.140119 | 10.1503/jpn.140119 | 46.4 | |||
| Neuropsychological mechanism underlying antidepressant effect: a systematic meta-analysis | 2015 | MDD | 50 | 1569 | https://www.ncbi.nlm.nih.gov/pubmed/24662929 | sci-hub.tw/10.1038/mp.2014.24 | sci-hub.la/10.1038/mp.2014.24 | 10.1038/mp.2014.24 | 31.4 | |||
| Molecular imaging of striatal dopamine transporters in major depression--a meta-analysis. | 2015 | MDD | 12 | 523 | https://www.ncbi.nlm.nih.gov/pubmed/25497470 | sci-hub.tw/10.1016/j.jad.2014.11.045 | sci-hub.la/10.1016/j.jad.2014.11.045 | 10.1016/j.jad.2014.11.045 | 43.6 | |||
| Meta-analysis of Functional Neuroimaging of Major Depressive Disorder in Youth. | 2015 | MDD | 14 | 520 | https://www.ncbi.nlm.nih.gov/pubmed/26332700 | sci-hub.tw/10.1001/jamapsychiatry.2015.1376 | sci-hub.la/10.1001/jamapsychiatry.2015.1376 | 10.1001/jamapsychiatry.2015.1376 | 37.1 | |||
| Optogenetics to study the circuits of fear- and depression-like behaviors: a critical analysis. | 2014 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/24727401 | sci-hub.tw/10.1016/j.pbb.2014.04.002 | sci-hub.la/10.1016/j.pbb.2014.04.002 | 10.1016/j.pbb.2014.04.002 | - | |||
| Deep brain stimulation of the human reward system for major depression--rationale, outcomes and outlook. | 2014 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/24513970 | sci-hub.tw/10.1038/npp.2014.28 | sci-hub.la/10.1038/npp.2014.28 | 10.1038/npp.2014.28 | - | |||
| Meta-analysis of molecular imaging of serotonin transporters in major depression. | 2014 | MDD | 18 | 736 | https://www.ncbi.nlm.nih.gov/pubmed/24802331 | sci-hub.tw/10.1038/jcbfm.2014.82 | sci-hub.la/10.1038/jcbfm.2014.82 | 10.1038/jcbfm.2014.82 | 40.9 | |||
| Optogenetic dissection of neural circuits underlying emotional valence and motivated behaviors. | 2013 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/23142759 | sci-hub.tw/10.1016/j.brainres.2012.11.001 | sci-hub.la/10.1016/j.brainres.2012.11.001 | 10.1016/j.brainres.2012.11.001 | - | |||
| The brain reward circuitry in mood disorders. | 2013 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/23942470 | sci-hub.tw/10.1038/nrn3381 | sci-hub.la/10.1038/nrn3381 | 10.1038/nrn3381 | - | |||
| Neuroreceptor imaging in depression. | 2013 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/22691454 | sci-hub.tw/10.1016/j.nbd.2012.06.001 | sci-hub.la/10.1016/j.nbd.2012.06.001 | 10.1016/j.nbd.2012.06.001 | - | |||
| Gray matter volume in major depressive disorder: a meta-analysis of voxel-based morphometry studies. | 2013 | MDD | 20 | 1594 | https://www.ncbi.nlm.nih.gov/pubmed/23146253 | sci-hub.tw/10.1016/j.pscychresns.2012.06.006 | sci-hub.la/10.1016/j.pscychresns.2012.06.006 | 10.1016/j.pscychresns.2012.06.006 | 79.7 | |||
| Meta-analytic evidence for neuroimaging models of depression: state or trait? | 2013 | MDD | 40 | 1270 | https://www.ncbi.nlm.nih.gov/pubmed/23890584 | sci-hub.tw/10.1016/j.jad.2013.07.002 | sci-hub.la/10.1016/j.jad.2013.07.002 | 10.1016/j.jad.2013.07.002 | 31.8 | |||
| Resting-state brain activity in schizophrenia and major depression: a quantitative meta-analysis. | 2013 | MDD | 11 | 470 | https://www.ncbi.nlm.nih.gov/pubmed/22080493 | sci-hub.tw/10.1093/schbul/sbr151 | sci-hub.la/10.1093/schbul/sbr151 | 10.1093/schbul/sbr151 | 42.7 | |||
| A meta-analysis of neurofunctional imaging studies of emotion and cognition in major depression. | 2012 | MDD | 40 | 1127 | https://www.ncbi.nlm.nih.gov/pubmed/22521254 | sci-hub.tw/10.1016/j.neuroimage.2012.04.005 | sci-hub.la/10.1016/j.neuroimage.2012.04.005 | 10.1016/j.neuroimage.2012.04.005 | 28.2 | |||
| Functional neuroimaging of major depressive disorder: a meta-analysis and new integration of base line activation and neural response data. | 2012 | MDD | 14 | 599 | https://www.ncbi.nlm.nih.gov/pubmed/22535198 | sci-hub.tw/10.1176/appi.ajp.2012.11071105 | sci-hub.la/10.1176/appi.ajp.2012.11071105 | 10.1176/appi.ajp.2012.11071105 | 42.8 | |||
| Structural and functional neuroimaging studies of the suicidal brain. | 2011 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/21216267 | sci-hub.tw/10.1016/j.pnpbp.2010.12.026 | sci-hub.la/10.1016/j.pnpbp.2010.12.026 | 10.1016/j.pnpbp.2010.12.026 | - | |||
| Structural neuroimaging studies in major depressive disorder. Meta-analysis and comparison with bipolar disorder. | 2011 | MDD | 143 | 18379 | https://www.ncbi.nlm.nih.gov/pubmed/21727252 | sci-hub.tw/10.1001/archgenpsychiatry.2011.60 | sci-hub.la/10.1001/archgenpsychiatry.2011.60 | 10.1001/archgenpsychiatry.2011.60 | 128.5 | |||
| Functional neuroimaging of reward processing and decision-making: a review of aberrant motivational and affective processing in addiction and mood disorders. | 2008 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/18675846 | sci-hub.tw/10.1016/j.brainresrev.2008.07.004 | sci-hub.la/10.1016/j.brainresrev.2008.07.004 | 10.1016/j.brainresrev.2008.07.004 | - | |||
| Dynamics of the dopaminergic system as a key component to the understanding of depression. | 2008 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/18772037 | sci-hub.tw/10.1016/S0079-6123(08)00913-8 | sci-hub.la/10.1016/S0079-6123(08)00913-8 | 10.1016/S0079-6123(08)00913-8 | - | |||
| The mesolimbic dopamine reward circuit in depression. | 2006 | MDD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/16566899 | sci-hub.tw/10.1016/j.biopsych.2005.09.018 | sci-hub.la/10.1016/j.biopsych.2005.09.018 | 10.1016/j.biopsych.2005.09.018 | - | |||
| Review of 1H magnetic resonance spectroscopy findings in major depressive disorder: a meta-analysis. | 2006 | MDD | 42 | 1688 | https://www.ncbi.nlm.nih.gov/pubmed/16806850 | sci-hub.tw/10.1016/j.pscychresns.2005.12.004 | sci-hub.la/10.1016/j.pscychresns.2005.12.004 | 10.1016/j.pscychresns.2005.12.004 | 40.2 | |||
| Distinct Subcortical Volume Alterations in Pediatric and Adult OCD: A Worldwide Meta- and Mega-Analysis. | 2017 | OCD | 1 | 3589 | https://www.ncbi.nlm.nih.gov/pubmed/27609241 | sci-hub.tw/10.1176/appi.ajp.2016.16020201 | sci-hub.la/10.1176/appi.ajp.2016.16020201 | 10.1176/appi.ajp.2016.16020201 | 3589.0 | |||
| Neural correlates of affective and non-affective cognition in obsessive compulsive disorder: A meta-analysis of functional imaging studies. | 2017 | OCD | 54 | 2345 | https://www.ncbi.nlm.nih.gov/pubmed/28992533 | sci-hub.tw/10.1016/j.eurpsy.2017.08.001 | sci-hub.la/10.1016/j.eurpsy.2017.08.001 | 10.1016/j.eurpsy.2017.08.001 | 43.4 | |||
| Meta-analytic investigations of common and distinct grey matter alterations in youths and adults with obsessive-compulsive disorder. | 2017 | OCD | 25 | 1575 | https://www.ncbi.nlm.nih.gov/pubmed/28442404 | sci-hub.tw/10.1016/j.neubiorev.2017.04.012 | sci-hub.la/10.1016/j.neubiorev.2017.04.012 | 10.1016/j.neubiorev.2017.04.012 | 63.0 | |||
| Structural and Functional Brain Abnormalities in Attention-Deficit/Hyperactivity Disorder and Obsessive-Compulsive Disorder: A Comparative Meta-analysis. | 2016 | OCD | 108 | 5274 | https://www.ncbi.nlm.nih.gov/pubmed/27276220 | sci-hub.tw/10.1001/jamapsychiatry.2016.0700 | sci-hub.la/10.1001/jamapsychiatry.2016.0700 | 10.1001/jamapsychiatry.2016.0700 | 48.8 | |||
| Meta-analytic investigations of structural grey matter, executive domain-related functional activations, and white matter diffusivity in obsessive compulsive disorder: an integrative review. | 2015 | OCD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/25766413 | sci-hub.tw/10.1016/j.neubiorev.2015.03.002 | sci-hub.la/10.1016/j.neubiorev.2015.03.002 | 10.1016/j.neubiorev.2015.03.002 | - | |||
| Multimodal voxel-based meta-analysis of white matter abnormalities in obsessive-compulsive disorder. | 2015 | OCD | 22 | 1112 | https://www.ncbi.nlm.nih.gov/pubmed/24407265 | sci-hub.tw/10.1038/npp.2014.5 | sci-hub.la/10.1038/npp.2014.5 | 10.1038/npp.2014.5 | 50.5 | |||
| Brain circuitries of obsessive compulsive disorder: a systematic review and meta-analysis of diffusion tensor imaging studies. | 2013 | OCD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/24177038 | sci-hub.tw/10.1016/j.neubiorev.2013.10.008 | sci-hub.la/10.1016/j.neubiorev.2013.10.008 | 10.1016/j.neubiorev.2013.10.008 | - | |||
| Neuroimaging of cognitive brain function in paediatric obsessive compulsive disorder: a review of literature and preliminary meta-analysis. | 2012 | OCD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/22678698 | sci-hub.tw/10.1007/s00702-012-0813-z | sci-hub.la/10.1007/s00702-012-0813-z | 10.1007/s00702-012-0813-z | - | |||
| Reduction of N-acetylaspartate in the medial prefrontal cortex correlated with symptom severity in obsessive-compulsive disorder: meta-analyses of (1)H-MRS studies. | 2012 | OCD | 16 | 458 | https://www.ncbi.nlm.nih.gov/pubmed/22892718 | sci-hub.tw/10.1038/tp.2012.78 | sci-hub.la/10.1038/tp.2012.78 | 10.1038/tp.2012.78 | 28.6 | |||
| Gray matter alterations in obsessive-compulsive disorder: an anatomic likelihood estimation meta-analysis. | 2010 | OCD | 10 | 661 | https://www.ncbi.nlm.nih.gov/pubmed/19890260 | sci-hub.tw/10.1038/npp.2009.175 | sci-hub.la/10.1038/npp.2009.175 | 10.1038/npp.2009.175 | 66.1 | |||
| Meta-analysis of brain volume changes in obsessive-compulsive disorder. | 2009 | OCD | 14 | 778 | https://www.ncbi.nlm.nih.gov/pubmed/18718575 | sci-hub.tw/10.1016/j.biopsych.2008.06.019 | sci-hub.la/10.1016/j.biopsych.2008.06.019 | 10.1016/j.biopsych.2008.06.019 | 55.6 | |||
| Voxel-wise meta-analysis of grey matter changes in obsessive-compulsive disorder. | 2009 | OCD | 12 | 777 | https://www.ncbi.nlm.nih.gov/pubmed/19880927 | sci-hub.tw/10.1192/bjp.bp.108.055046 | sci-hub.la/10.1192/bjp.bp.108.055046 | 10.1192/bjp.bp.108.055046 | 64.8 | |||
| Integrating evidence from neuroimaging and neuropsychological studies of obsessive-compulsive disorder: the orbitofronto-striatal model revisited. | 2008 | OCD | - | - | https://www.ncbi.nlm.nih.gov/pubmed/18061263 | sci-hub.tw/10.1016/j.neubiorev.2007.09.005 | sci-hub.la/10.1016/j.neubiorev.2007.09.005 | 10.1016/j.neubiorev.2007.09.005 | - | |||
| Provocation of obsessive–compulsive symptoms: a quantitative voxel-based meta-analysis of functional neuroimaging studies | 2008 | OCD | 8 | 94 | https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2527721/ | sci-hub.tw/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2527721/ | sci-hub.la/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2527721/ | https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2527721/ | 11.8 | |||
| A meta-analysis of functional neuroimaging in obsessive-compulsive disorder. | 2004 | OCD | 13 | 40 | https://www.ncbi.nlm.nih.gov/pubmed/15546704 | sci-hub.tw/10.1016/j.pscychresns.2004.07.001 | sci-hub.la/10.1016/j.pscychresns.2004.07.001 | 10.1016/j.pscychresns.2004.07.001 | 3.1 | |||
| Different patterns of 5-HT receptor and transporter dysfunction in neuropsychiatric disorders--a comparative analysis of in vivo imaging findings. | 2016 | PSY | 136 | - | https://www.ncbi.nlm.nih.gov/pubmed/26376220 | sci-hub.tw/10.1515/revneuro-2015-0014 | sci-hub.la/10.1515/revneuro-2015-0014 | 10.1515/revneuro-2015-0014 | - | |||
| Mapping anhedonia-specific dysfunction in a transdiagnostic approach: an ALE meta-analysis. | 2016 | PSY | 89 | 3272 | https://www.ncbi.nlm.nih.gov/pubmed/26487590 | sci-hub.tw/10.1007/s11682-015-9457-6 | sci-hub.la/10.1007/s11682-015-9457-6 | 10.1007/s11682-015-9457-6 | 36.8 | |||
| The serotonin transporter in psychiatric disorders: insights from PET imaging. | 2015 | PSY | - | - | https://www.ncbi.nlm.nih.gov/pubmed/26249305 | sci-hub.tw/10.1016/S2215-0366(15)00232-1 | sci-hub.la/10.1016/S2215-0366(15)00232-1 | 10.1016/S2215-0366(15)00232-1 | - | |||
| Mechanisms Underlying Motivational Deficits in Psychopathology: Similarities and Differences in Depression and Schizophrenia. | 2014 | PSY | - | - | https://www.ncbi.nlm.nih.gov/pubmed/26026289 | sci-hub.tw/10.1007/7854_2015_376 | sci-hub.la/10.1007/7854_2015_376 | 10.1007/7854_2015_376 | - | |||
| Brain imaging findings in children and adolescents with mental disorders: a cross-sectional review | 2010 | PSY | 274 | - | https://www.ncbi.nlm.nih.gov/pubmed/20620025 | sci-hub.tw/10.1016/j.eurpsy.2010.04.010 | sci-hub.la/10.1016/j.eurpsy.2010.04.010 | 10.1016/j.eurpsy.2010.04.010 | - | |||
| The IMAGEN study: reinforcement-related behaviour in normal brain function and psychopathology. | 2010 | PSY | - | - | https://www.ncbi.nlm.nih.gov/pubmed/21102431 | sci-hub.tw/10.1038/mp.2010.4 | sci-hub.la/10.1038/mp.2010.4 | 10.1038/mp.2010.4 | - | |||
| Aberrant Resting-State Brain Activity in Post-Traumatic Stress Disorder: A Meta-Analysis And Systematic Review. | 2016 | PTSD | 23 | 663 | https://www.ncbi.nlm.nih.gov/pubmed/26918313 | sci-hub.tw/10.1002/da.22478 | sci-hub.la/10.1002/da.22478 | 10.1002/da.22478 | 28.8 | |||
| In search of the trauma memory: a meta-analysis of functional neuroimaging studies of symptom provocation in posttraumatic stress disorder (PTSD). | 2013 | PTSD | 14 | 66 | https://www.ncbi.nlm.nih.gov/pubmed/23536785 | sci-hub.tw/10.1371/journal.pone.0058150 | sci-hub.la/10.1371/journal.pone.0058150 | 10.1371/journal.pone.0058150 | 4.7 | |||
| Common and distinct neural correlates of facial emotion processing in social anxiety disorder and Williams syndrome: A systematic review and voxel-based meta-analysis of functional resonance imaging studies. | 2014 | SAD | 17 | 575 | https://www.ncbi.nlm.nih.gov/pubmed/25194208 | sci-hub.tw/10.1016/j.neuropsychologia.2014.08.027 | sci-hub.la/10.1016/j.neuropsychologia.2014.08.027 | 10.1016/j.neuropsychologia.2014.08.027 | 33.8 | |||
| Neuroimaging in social anxiety disorder—a meta-analytic review resulting in a new neurofunctional model. | 2014 | SAD | 19 | 409 | https://www.ncbi.nlm.nih.gov/pubmed/25124509 | sci-hub.tw/10.1016/j.neubiorev.2014.08.003 | sci-hub.la/10.1016/j.neubiorev.2014.08.003 | 10.1016/j.neubiorev.2014.08.003 | 21.5 | |||
| Schizophrenia symptomatic associations with diffusion tensor imaging measured fractional anisotropy of brain: a meta-analysis. | 2017 | SCZ | 33 | 2242 | https://www.ncbi.nlm.nih.gov/pubmed/28550466 | sci-hub.tw/10.1007/s00234-017-1844-9 | sci-hub.la/10.1007/s00234-017-1844-9 | 10.1007/s00234-017-1844-9 | 67.9 | |||
| Positive symptoms associate with cortical thinning in the superior temporal gyrus via the ENIGMA Schizophrenia consortium. | 2017 | SCZ | 1 | 1987 | https://www.ncbi.nlm.nih.gov/pubmed/28369804 | sci-hub.tw/10.1111/acps.12718 | sci-hub.la/10.1111/acps.12718 | 10.1111/acps.12718 | 1987.0 | |||
| Patients with schizophrenia show aberrant patterns of basal ganglia activation: Evidence from ALE meta-analysis. | 2017 | SCZ | 42 | 1290 | https://www.ncbi.nlm.nih.gov/pubmed/28275545 | sci-hub.tw/10.1016/j.nicl.2017.01.034 | sci-hub.la/10.1016/j.nicl.2017.01.034 | 10.1016/j.nicl.2017.01.034 | 30.7 | |||
| Brain-Wide Analysis of Functional Connectivity in First-Episode and Chronic Stages of Schizophrenia. | 2017 | SCZ | 6 | 789 | https://www.ncbi.nlm.nih.gov/pubmed/27445261 | sci-hub.tw/10.1093/schbul/sbw099 | sci-hub.la/10.1093/schbul/sbw099 | 10.1093/schbul/sbw099 | 131.5 | |||
| Altered Hub Functioning and Compensatory Activations in the Connectome: A Meta-Analysis of Functional Neuroimaging Studies in Schizophrenia. | 2016 | SCZ | 314 | 10000 | https://www.ncbi.nlm.nih.gov/pubmed/26472684 | sci-hub.tw/10.1093/schbul/sbv146 | sci-hub.la/10.1093/schbul/sbv146 | 10.1093/schbul/sbv146 | 31.8 | |||
| The neural mechanisms of hallucinations: A quantitative meta-analysis of neuroimaging studies. | 2016 | SCZ | 17 | 211 | https://www.ncbi.nlm.nih.gov/pubmed/27473935 | sci-hub.tw/10.1016/j.neubiorev.2016.05.037 | sci-hub.la/10.1016/j.neubiorev.2016.05.037 | 10.1016/j.neubiorev.2016.05.037 | 12.4 | |||
| Rostral medial prefrontal dysfunctions and consummatory pleasure in schizophrenia: a meta-analysis of functional imaging studies. | 2015 | SCZ | 19 | 615 | https://www.ncbi.nlm.nih.gov/pubmed/25637357 | sci-hub.tw/10.1016/j.pscychresns.2015.01.001 | sci-hub.la/10.1016/j.pscychresns.2015.01.001 | 10.1016/j.pscychresns.2015.01.001 | 32.4 | |||
| Alterations in the serotonin system in schizophrenia: a systematic review and meta-analysis of postmortem and molecular imaging studies. | 2014 | SCZ | - | - | https://www.ncbi.nlm.nih.gov/pubmed/24971825 | sci-hub.tw/10.1016/j.neubiorev.2014.06.005 | sci-hub.la/10.1016/j.neubiorev.2014.06.005 | 10.1016/j.neubiorev.2014.06.005 | - | |||
| Alterations in cortical and extrastriatal subcortical dopamine function in schizophrenia: systematic review and meta-analysis of imaging studies. | 2014 | SCZ | 23 | 543 | https://www.ncbi.nlm.nih.gov/pubmed/25029687 | sci-hub.tw/10.1192/bjp.bp.113.132308 | sci-hub.la/10.1192/bjp.bp.113.132308 | 10.1192/bjp.bp.113.132308 | 23.6 | |||
| Brain vs behavior: an effect size comparison of neuroimaging and cognitive studies of genetic risk for schizophrenia. | 2013 | SCZ | - | - | https://www.ncbi.nlm.nih.gov/pubmed/22499782 | sci-hub.tw/10.1093/schbul/sbs056 | sci-hub.la/10.1093/schbul/sbs056 | 10.1093/schbul/sbs056 | - | |||
| Striatal presynaptic dopamine in schizophrenia, Part I: meta-analysis of dopamine active transporter (DAT) density. | 2013 | SCZ | 13 | 349 | https://www.ncbi.nlm.nih.gov/pubmed/22282456 | sci-hub.tw/10.1093/schbul/sbr111 | sci-hub.la/10.1093/schbul/sbr111 | 10.1093/schbul/sbr111 | 26.8 | |||
| Striatal presynaptic dopamine in schizophrenia, part II: meta-analysis of [(18)F/(11)C]-DOPA PET studies. | 2013 | SCZ | 11 | 244 | https://www.ncbi.nlm.nih.gov/pubmed/22282454 | sci-hub.tw/10.1093/schbul/sbr180 | sci-hub.la/10.1093/schbul/sbr180 | 10.1093/schbul/sbr180 | 22.2 | |||
| Meta-analysis of functional neuroimaging studies of emotion perception and experience in schizophrenia. | 2012 | SCZ | 26 | 872 | https://www.ncbi.nlm.nih.gov/pubmed/21993193 | sci-hub.tw/10.1016/j.biopsych.2011.09.007 | sci-hub.la/10.1016/j.biopsych.2011.09.007 | 10.1016/j.biopsych.2011.09.007 | 33.5 | |||
| Quantitative meta-analysis on state and trait aspects of auditory verbal hallucinations in schizophrenia. | 2012 | SCZ | 15 | 274 | https://www.ncbi.nlm.nih.gov/pubmed/21177743 | sci-hub.tw/10.1093/schbul/sbq152 | sci-hub.la/10.1093/schbul/sbq152 | 10.1093/schbul/sbq152 | 18.3 | |||
| The "paradoxical" engagement of the primary auditory cortex in patients with auditory verbal hallucinations: a meta-analysis of functional neuroimaging studies. | 2011 | SCZ | 12 | 477 | https://www.ncbi.nlm.nih.gov/pubmed/21872614 | sci-hub.tw/10.1016/j.neuropsychologia.2011.08.010 | sci-hub.la/10.1016/j.neuropsychologia.2011.08.010 | 10.1016/j.neuropsychologia.2011.08.010 | 39.8 | |||
| Facial emotion processing in schizophrenia: a meta-analysis of functional neuroimaging data. | 2010 | SCZ | 17 | - | https://www.ncbi.nlm.nih.gov/pubmed/19336391 | sci-hub.tw/10.1093/schbul/sbn190 | sci-hub.la/10.1093/schbul/sbn190 | 10.1093/schbul/sbn190 | - | |||
| Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. | 2009 | SCZ | 41 | - | https://www.ncbi.nlm.nih.gov/pubmed/19652121 | sci-hub.tw/10.1001/archgenpsychiatry.2009.91 | sci-hub.la/10.1001/archgenpsychiatry.2009.91 | 10.1001/archgenpsychiatry.2009.91 | - | |||
| Neural correlates of somatoform disorders from a meta-analytic perspective on neuroimaging studies. | 2016 | SD | 10 | 447 | https://www.ncbi.nlm.nih.gov/pubmed/27182487 | sci-hub.tw/10.1016/j.nicl.2016.04.001 | sci-hub.la/10.1016/j.nicl.2016.04.001 | 10.1016/j.nicl.2016.04.001 | 44.7 | |||
| Neural correlates of conversion disorder: overview and meta-analysis of neuroimaging studies on motor conversion disorder. | 2016 | SD | 12 | 335 | https://www.ncbi.nlm.nih.gov/pubmed/27283002 | sci-hub.tw/10.1186/s12888-016-0890-x | sci-hub.la/10.1186/s12888-016-0890-x | 10.1186/s12888-016-0890-x | 27.9 |
| Title | Summary | Pubmed |
|---|---|---|
| Melancholy, anhedonia, apathy: the search for separable behaviors and neural circuits in depression. | In rodent models of depression, activity in the mPFC as a whole relates to reduced sucrose preference and sociability, with are thought to be analogous to anhedonia and social withdrawal, respectively, in humans. Inhibiting this region via stimulating it at a supraphysiological frequency appears to attenuate these deficits. | https://www.ncbi.nlm.nih.gov/pubmed/29529482 |
| Circuit-based frameworks of depressive behaviors: The role of reward circuitry and beyond. | Evidence related to the role of the VTA in producing depression-like behaviors in animals is complex. | https://www.ncbi.nlm.nih.gov/pubmed/29309799 |
| Treatment resistant depression: A multi-scale, systems biology approach. | https://www.ncbi.nlm.nih.gov/pubmed/28859997 | |
| Dopamine System Dysregulation in Major Depressive Disorders. | A | https://www.ncbi.nlm.nih.gov/pubmed/29106542 |
| Progress in understanding mood disorders: optogenetic dissection of neural circuits. | A | https://www.ncbi.nlm.nih.gov/pubmed/23682971 |
| Presentation and Neurobiology of Anhedonia in Mood Disorders: Commonalities and Distinctions. | A | https://www.ncbi.nlm.nih.gov/pubmed/29520717 |
| Neuroimaging genomic studies in major depressive disorder: A systematic review. | A | https://www.ncbi.nlm.nih.gov/pubmed/29476595 |
| Lateral habenula in the pathophysiology of depression. | A | https://www.ncbi.nlm.nih.gov/pubmed/29175713 |
| Intrinsic cerebral activity at resting state in adults with major depressive disorder: A meta-analysis. | A | https://www.ncbi.nlm.nih.gov/pubmed/28174129 |
| Characterization of brain blood flow and the amplitude of low-frequency fluctuations in major depressive disorder: A multimodal meta-analysis. | A | https://www.ncbi.nlm.nih.gov/pubmed/28068619 |
| Altered Brain Activity in Unipolar Depression Revisited: Meta-analyses of Neuroimaging Studies. | A | https://www.ncbi.nlm.nih.gov/pubmed/27829086 |
The cause and mechanism of schizophrenia is unknown. Evidence from phenomenology, pharmacology, neuroimaging, post mortem studies, genetics, and animal models implicate a number of possible and likely interrelated mechanisms, such as abnormalities in dopaminergic signalling, glutaminergic neurotransmission, and neurodevelopment. Many frameworks have been hypothesized to link these biological abnormalities to symptoms, including psychological and computational theories.[52][53]
Abnormal dopamine signalling has been implicated in schizophrenia by the efficacy of D2 receptor antagonists, as well as the consistent observation in positron emission tomography of elevated dopamine synthesis[54] and release during acute psychosis.[55] Abnormalities in dopaminergic symptoms have been hypothesized to underlie delusions via dysfunctional signalling of salience.[56][57][58] Dopaminergic predictions errors, which mediate learning when expectancies are violated, are abnormal in schizophrenia, and these abnormalities correlate with the severity of delusions. Furthermore, impaired learning, putatively reflecting the functionality of the dopaminergic system, is present in schizophrenia and correlates with delusion severity.[59] Dysfunctional prediction errors may be related to hyperactive input from the hippocampus, which is observed to be metabolically overactive in schizophrenia,[56] and in turn may be related to abnormalities in NMDA receptor functioning on hippocampal interneurons.[60] Hypoactivation of D1 receptors in the prefrontal cortex may also be responsible for deficits in working memory and cognition,[61][62][63][64] although direct evidence from neuroreceptor imaging studies is inconsistent.[65]
Reduced NMDA receptor signalling is suggested by multiple lines of evidence. Post-mortem studies demonstrate reduced NMDA receptor expression and NMDA receptor antagonists mimic both schizophrenia symptoms and the electrophysiological abnormalities associated with schizophrenia (notable reduced mismatch negativity and P300).[66][67][68] Two meta analyses of magnetic resonance spectroscopy studies have found evidence interpreted to be consistent with abnormal glutaminergic signalling, possible involving NMDA receptor abnormalities, despite contradictory findings (i.e. reduced versus elevated glutamate/glutamine ratios).[69][70] This deficit in NMDA signalling may be related to the abnormalities observed in parvalbumin interneurons that express NMDA receptors.[71] Post mortem studies consistently find that a subset of these neurons fail to express GAD67 in addition to abnormalities in morphology,[72] although neuroimaging studies examining indicators of GABAergic signalling do not consistently report abnormalities.[73][74] The subsets of interneurons that are abnormal in schizophrenia are responsible for the synchronizing of neural ensembles that is necessary during working memory tasks, a process that is electrophysiologically reflected in gamma frequency (30-80 Hz) oscillations. Both working memory tasks and gamma oscillations are impaired in schizophrenia, which may reflect abnormal interneuron functionality.[75][76][77][78][79]
Multiple lines of evidence suggest that schizophrenia has a neurodevelopmental component.[80] Schizophrenia is associated with premorbid impairments in cognition, social functioning, and motor skills,[81] and prenatal insults such as maternal infection,[82][83] maternal malnutrition and obsteric complications all increase risk for schizophrenia.[84] Animal models of these insults demonstrate patterns of cellular and molecular abnormalities similar to those in schizophrenia, such as increased RELN methylation and abnormal GABAergic cell development.[85] Schizophrenia usually emerges symptomatically during late adolescence, 18-25, an age period that overlaps with certain stages of neurodevelopment that are implicated in schizophrenia, such as abnormal synaptic pruning and myelination.[86]
Deficits in executive functions, such as planning, inhibition, and working memory, are pervasive in schizophrenia. Although these functions are dissociable, their dysfunction in schizophrenia may reflect an underlying deficit in the ability to represent goal related information in working memory, and to utilize this to direct cognition and behavior.[87][88] These impairments have been linked to a number of neuroimaging and neuropathological abnormalities. For example, functional neuroimaging studies report evidence of reduced neural processing efficiency, whereby the dorsolateral prefrontal cortex is activated to a greater degree to achieve a certain level of performance relative to controls on working memory tasks. These abnormalities may be linked to the consistent post-mortem finding of reduced neuropil, evidenced by increased pyramidal cell density, reduced dentritic spine density,[89] and reduced expression mRNA associated with synapses.[90] These cellular and functional abnormalities may also be reflected in structural neuroimaging studies that find reduced grey matter volume in association with deficits in working memory tasks.[89]
Different symptoms have been linked to specific neuroanatomical abnormalities or neurobiological models. For example, positive and negative symptoms have been linked to reduced cortical thickness in the superior temporal lobe,[91] and orbitofrontal cortex, respectively.[92] Auditory hallucinations, a prominent component of positive symptoms, are also reflected in functional hyperactivity of auditory cortices, and in the predictive coding framework are hypothesized to reflect impaired feedforward cancellation of internally generated speech.[93] Anhedonia, traditionally defined as a reduced capacity to experience pleasure, is frequently reported in schizophrenia. However, a large body of evidence suggests that hedonic responses are intact in schizophrenia,[94] and that what is reported to be anhedonia is a reflection of dysfunction in other processes related to reward.[95] Overall, a failure of online maintenance and reward associativity is thought to lead to impairment in the generation of cognition and behavior required to obtain rewards, despite normal hedonic responses.[96] A meta analysis of neuroimaging studies examining reward related paradigms reported results consistent with impairments in the neural substrates that mediate learning, but not in the experience of reward.[97]
Bayesian models of brain function have been utilized to link abnormalities in cellular functioning to symptoms. These models propose that the brain generates predictive models in order to explain sensory information, and that predictive units are organized in a hierarchical fashion that reflect a more abstract model of causes for sensory information higher in the hierarchy. The essential computation performed by these models involves the minimization of error generated by discrepancies in top down predictions by modifying synaptic weights with the least precision.[98][99] Improper specification of precision in bayesian models is largely consistent with the behavioral, neuroimaging, and electrophysiological abnormalities associated with schizophrenia. Both a failure to attenuate sensory precision and excessive weighting or priors have been proposed as potential and not necessarily conflicting explanations.[100] In canonical models of circuits that mediate predictive coding, hypoactive NMDA receptor activation and abnormalities in dopaminergic signalling,[101] similar to that seen in schizophrenia, could theoretically result in classic symptoms of schizophrenia such as delusions and hallucinations.[102][59][103]



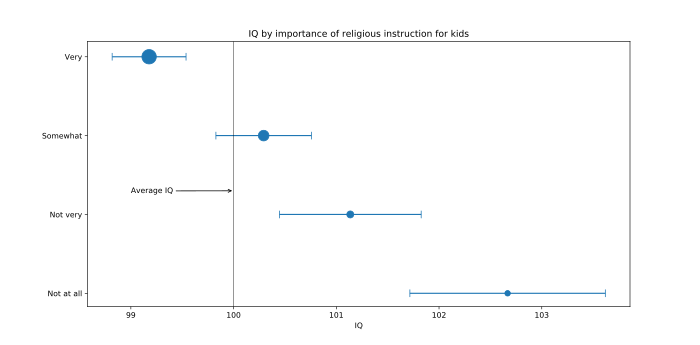
OLS Regression Results
==============================================================================
Dep. Variable: g factor R-squared: 0.202
Model: OLS Adj. R-squared: 0.201
Method: Least Squares F-statistic: 293.2
Date: Thu, 02 Aug 2018 Prob (F-statistic): 1.11e-169
Time: 12:11:03 Log-Likelihood: -4542.1
No. Observations: 3478 AIC: 9090.
Df Residuals: 3475 BIC: 9109.
Df Model: 3
Covariance Type: nonrobust
==============================================================================
coef std err t P>|t| [0.025 0.975]
------------------------------------------------------------------------------
RELIMP -0.0836 0.016 -5.372 0.000 -0.114 -0.053
AGE -0.4253 0.015 -27.717 0.000 -0.455 -0.395
GENDER -0.0582 0.015 -3.782 0.000 -0.088 -0.028
==============================================================================
Omnibus: 12.035 Durbin-Watson: 1.977
Prob(Omnibus): 0.002 Jarque-Bera (JB): 9.991
Skew: 0.057 Prob(JB): 0.00677
Kurtosis: 2.763 Cond. No. 1.26
==============================================================================
OLS Regression Results
==============================================================================
Dep. Variable: g factor R-squared: 0.199
Model: OLS Adj. R-squared: 0.198
Method: Least Squares F-statistic: 288.0
Date: Thu, 02 Aug 2018 Prob (F-statistic): 5.32e-167
Time: 12:11:03 Log-Likelihood: -4566.8
No. Observations: 3491 AIC: 9140.
Df Residuals: 3488 BIC: 9158.
Df Model: 3
Covariance Type: nonrobust
==============================================================================
coef std err t P>|t| [0.025 0.975]
------------------------------------------------------------------------------
PRAY -0.0586 0.016 -3.718 0.000 -0.090 -0.028
AGE -0.4313 0.015 -28.260 0.000 -0.461 -0.401
GENDER -0.0600 0.016 -3.820 0.000 -0.091 -0.029
==============================================================================
Omnibus: 11.895 Durbin-Watson: 1.976
Prob(Omnibus): 0.003 Jarque-Bera (JB): 9.650
Skew: 0.043 Prob(JB): 0.00803
Kurtosis: 2.757 Cond. No. 1.33
==============================================================================
OLS Regression Results
==============================================================================
Dep. Variable: g factor R-squared: 0.201
Model: OLS Adj. R-squared: 0.201
Method: Least Squares F-statistic: 293.0
Date: Thu, 02 Aug 2018 Prob (F-statistic): 1.27e-169
Time: 12:11:03 Log-Likelihood: -4555.1
No. Observations: 3487 AIC: 9116.
Df Residuals: 3484 BIC: 9135.
Df Model: 3
Covariance Type: nonrobust
==============================================================================
coef std err t P>|t| [0.025 0.975]
------------------------------------------------------------------------------
REL -0.0780 0.015 -5.050 0.000 -0.108 -0.048
AGE -0.4275 0.015 -27.954 0.000 -0.457 -0.398
GENDER -0.0622 0.015 -4.062 0.000 -0.092 -0.032
==============================================================================
Omnibus: 11.538 Durbin-Watson: 1.967
Prob(Omnibus): 0.003 Jarque-Bera (JB): 9.534
Skew: 0.050 Prob(JB): 0.00851
Kurtosis: 2.764 Cond. No. 1.22
==============================================================================
REL: Response to "How religious are you?", on a scale of 1-4, with 1 being the least religious and 4 being the most religious RELIMP: Response to "How important is religion to you?", on a scale of 1-4, with 1 being the least important and 4 being the most important PRAY: Response to "How often do you pray?", on a scale of 1-6, with 1 being never and 6 being once or more a day