 | |
| Clinical data | |
|---|---|
| Trade names | Ztalmy |
| Other names | GNX; CCD-1042; 3β-Methyl-5α-pregnan-3α-ol-20-one; 3α-Hydroxy-3β-methyl-5α-pregnan-20-one |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Neurosteroid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.210.937 |
| Chemical and physical data | |
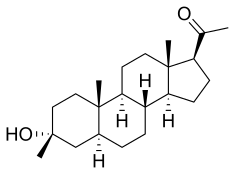
| Formula | C22H36O2 |
| Molar mass | 332.528 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ganaxolone, sold under the brand name Ztalmy, is a medication used to treat seizures in people with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder.[1][3] Ganaxolone is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive modulator.[1]
The most common side effects of treatment with ganaxolone include somnolence (sleepiness), fever, excessive saliva or drooling, and seasonal allergy.[4]
Ganaxolone was approved for medical use in the United States in March 2022,[1][4] and in the European Union in July 2023.[2] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication.[5][6]
Medical uses
[edit]Ganaxolone is indicated for the treatment of seizures associated with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder.[1][2]
Pharmacology
[edit]Mechanism of action
[edit]The exact mechanism of action for ganaxolone is unknown; however, results from animal studies suggest that it acts by blocking seizure propagation and elevating seizure thresholds.[7][8]
Ganaxolone is thought to modulate both synaptic and extrasynaptic GABAA receptors to normalize over-excited neurons.[3] Ganaxolone's activation of the extrasynaptic receptor is an additional mechanism that provides stabilizing effects that potentially differentiates it from other drugs that increase GABA signaling.[3]
Ganaxolone binds to allosteric sites of the GABAA receptor to modulate and open the chloride ion channel, resulting in a hyperpolarization of the neuron.[3] This causes an inhibitory effect on neurotransmission, reducing the chance of a successful action potential (depolarization) from occurring.[3][7][8]
It is unknown whether ganaxolone possesses significant hormonal activity in vivo, with a 2020 study finding evidence of in vitro binding to the membrane progesterone receptor.[9]
Chemistry
[edit]Ganaxolone is an analog of the neurosteroid allopregnanolone that possesses no known hormonal activity and, instead, is thought to primarily function by binding to GABAA receptors as a positive allosteric modulator.[10]
Other pregnane neurosteroids include alfadolone, alfaxolone, hydroxydione, minaxolone, pregnanolone (eltanolone), and renanolone, among others.[11]
History
[edit]The FDA approved ganaxolone based on evidence from a single, double-blind, randomized, placebo-controlled study (Study 1, NCT03572933) of 101 participants with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder who were two years of age and older.[4] The trial was conducted at 36 sites in 8 countries including Australia, France, Israel, Italy, Poland, Russian Federation, the United Kingdom, and the United States.[4] Forty-four (40.7%) of the participants were from US sites.[4] Safety was assessed from a pool of two clinical studies.[4] These include the study of participants with cyclin-dependent kinase-like 5 deficiency disorder and a clinical study that included seven additional participants from a trial of ganaxolone in children and young adults.[4]
References
[edit]- ^ a b c d e "Ztalmy- ganaxolone suspension". DailyMed. 15 November 2022. Archived from the original on 21 January 2023. Retrieved 21 January 2023.
- ^ a b c "Ztalmy EPAR". European Medicines Agency. 31 July 2023. Archived from the original on 25 August 2023. Retrieved 25 August 2023.
- ^ a b c d e Carter RB, Wood PL, Wieland S, Hawkinson JE, Belelli D, Lambert JJ, et al. (March 1997). "Characterization of the anticonvulsant properties of ganaxolone (CCD 1042; 3alpha-hydroxy-3beta-methyl-5alpha-pregnan-20-one), a selective, high-affinity, steroid modulator of the gamma-aminobutyric acid(A) receptor". The Journal of Pharmacology and Experimental Therapeutics. 280 (3): 1284–1295. PMID 9067315.
- ^ a b c d e f g "Drug Trials Snapshots: Ztalmy". U.S. Food and Drug Administration. 18 March 2022. Retrieved 9 October 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Advancing Health Through Innovation: New Drug Therapy Approvals 2022". U.S. Food and Drug Administration (FDA). 10 January 2023. Archived from the original on 21 January 2023. Retrieved 22 January 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ New Drug Therapy Approvals 2022 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 14 January 2024. Retrieved 14 January 2024.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b Kaminski RM, Livingood MR, Rogawski MA (July 2004). "Allopregnanolone analogs that positively modulate GABA receptors protect against partial seizures induced by 6-Hz electrical stimulation in mice". Epilepsia. 45 (7): 864–867. doi:10.1111/j.0013-9580.2004.04504.x. PMID 15230714. S2CID 21974013.
- ^ a b Reddy DS, Rogawski MA (May 2010). "Ganaxolone suppression of behavioral and electrographic seizures in the mouse amygdala kindling model". Epilepsy Research. 89 (2–3): 254–260. doi:10.1016/j.eplepsyres.2010.01.009. PMC 2854307. PMID 20172694.
- ^ Thomas P, Pang Y (24 June 2020). "Anti-apoptotic Actions of Allopregnanolone and Ganaxolone Mediated Through Membrane Progesterone Receptors (PAQRs) in Neuronal Cells". Frontiers in Endocrinology. 11 (417): 417. doi:10.3389/fendo.2020.00417. PMC 7331777. PMID 32670200.
- ^ "PubChem compound summary for ganaxolone". PubChem databade. National Library of Medicine (National Center for Biotechnology Information). Archived from the original on 10 December 2022. Retrieved 6 August 2022.
- ^ US20190160078A1, Masuoka, Lorianne K. & Lappalainen, Jaakko, "Ganaxolone for use in treating genetic epileptic disorders", issued 2019-05-30
External links
[edit]- Clinical trial number NCT03572933 for "Study of Adjunctive Ganaxolone Treatment in Children and Young Adults With CDKL5 Deficiency Disorder (Marigold)" at ClinicalTrials.gov
| GABAergics | |||||
|---|---|---|---|---|---|
| Channel modulators | |||||
| Others |
| ||||
| |||||