 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /sɛmˈæɡlʊtaɪd/ sem-AG-luu-tyde or /ˌsɛməˈɡluːtaɪd/ SEM-ə-GLOO-tyde |
| Trade names | Ozempic, Rybelsus, Wegovy, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618008 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Subcutaneous, oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 89% |
| Metabolism | Proteolysis |
| Elimination half-life | 7 days |
| Duration of action | 63.6 h |
| Excretion | Urine and feces |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ECHA InfoCard | 100.219.541 |
| Chemical and physical data | |
| Formula | C187H291N45O59 |
| Molar mass | 4113.641 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Semaglutide is an antidiabetic medication used for the treatment of type 2 diabetes and an anti-obesity medication used for long-term weight management.[19][20][21] It is a peptide similar to the hormone glucagon-like peptide-1 (GLP-1), modified with a side chain.[22][23] It can be administered by subcutaneous injection or taken orally.[24][13][14][15] It is sold under the brand names Ozempic[13] and Rybelsus[14] for diabetes, and under the brand name Wegovy for weight loss.[15]
Semaglutide is a glucagon-like peptide-1 receptor agonist.[13][14][15] The most common side effects include nausea, vomiting, diarrhea, abdominal pain, and constipation.[13][16][17][18][25]
It was approved for medical use in the US in 2017.[13][26] In 2021, it was the 90th most commonly prescribed medication in the United States, with more than 8 million prescriptions.[27][28]
Medical uses
[edit]Semaglutide is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes.[13][14]
The higher-dose formulation of semaglutide is indicated as an adjunct to diet and exercise for long-term weight management in adults with obesity (initial body mass index (BMI) ≥ 30 kg/m2) or who are overweight (initial BMI ≥ 27 kg/m2) and have at least one weight-related comorbidity.[15][18][29]
In March 2024, the US Food and Drug Administration (FDA) expanded the indication for semaglutide (Wegovy), in combination with a reduced calorie diet and increased physical activity, to reduce the risk of cardiovascular death, heart attack and stroke in obese or overweight adults with cardiovascular disease.[30]
Side effects
[edit]Possible adverse effects include nausea, diarrhea, vomiting, constipation, abdominal pain, headache, fatigue, indigestion/heartburn, dizziness, abdominal distension, belching, hypoglycemia (low blood glucose) in people with type 2 diabetes, flatulence, gastroenteritis, and gastroesophageal reflux disease (GERD).[15] It can also cause pancreatitis, gastroparesis, and bowel obstruction.[31]
The US FDA label for semaglutide contains a boxed warning for thyroid C-cell tumors in rodents.[13][14][15][25] It is unknown whether semaglutide causes thyroid C-cell tumors, including medullary thyroid carcinoma, in humans.[13][14][15][25][32]
Contraindications
[edit]Data from rodent studies of GLP-1-mediated thyroid C-cell hyperplasia[33] indicates that use is contraindicated in people with a personal or family history of medullary thyroid carcinoma or with multiple endocrine neoplasia type 2.[14][13]
Mechanism of action
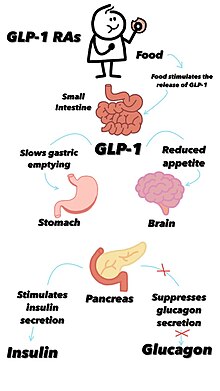
[edit]Semaglutide is a glucagon-like peptide-1 receptor agonist.[13][14][15] The drug decreases blood sugar levels. The decrease is theorized to be caused by the mimicking of the incretin glucagon-like peptide-1 (GLP-1).[34] It also appears to enhance growth of pancreatic beta cells, which are responsible for insulin production and release.[23][35] Additionally, it inhibits the production of glucagon, the hormone that increases glycogenolysis (release of stored carbohydrate from the liver) and gluconeogenesis (synthesis of new glucose). It reduces food intake by lowering appetite and slowing down digestion in the stomach,[22] helping reduce body weight.[36][37]
Structure and pharmacology
[edit]
Semaglutide is chemically similar to human GLP-1.[38] The first six amino acids of GLP-1 are missing.[38] Substitutions are made at GLP positions 8 and 34 (semaglutide positions 2 and 28), where alanine and lysine are replaced by 2-aminoisobutyric acid and arginine, respectively.[38] The substitution of the alanine prevents chemical breakdown by dipeptidyl peptidase-4.[39] The lysine at GLP position 26 (semaglutide position 20) has a long chain attached, ending with a chain of 17 carbon atoms and a carboxyl group.[39] This increases the drug's binding to blood protein (albumin), which enables longer presence in the blood circulation.[39]
Semaglutide's half-life in the blood is about seven days (165–184 hours).[23][40]
History
[edit]In the 1970s, Jens Juul Holst and Joel Habener began research on the GLP-1 hormone, initially in relation to duodenal ulcer disease.[41] They were examining hormones secreted during eating, and testing them on pig pancreases, leading to the discovery of GLP-1's significant potency in 1988. Their work, which later contributed significantly to diabetes and obesity treatments, earned them and Daniel Drucker the 2021 Warren Alpert Foundation Prize.[41] Research continued and in 1993 Michael Nauck managed to infuse GLP-1 into people with type 2 diabetes, stimulating insulin while inhibiting glucagon and bringing blood glucose to normal levels. However, treating diabetes patients with GLP-1 hormones resulted in significant side effects, leading researchers financed by Novo Nordisk to start looking to develop a suitable compound for therapeutic use.[41]
In 1998 a team of researchers at Novo Nordisk led by the scientist Lotte Bjerre Knudsen developed liraglutide, a glucagon-like peptide-1 receptor agonist that could be used to treat diabetes.[42] In June 2008, a phase II clinical trial began studying semaglutide, a once-weekly diabetes therapy as a longer-acting alternative to liraglutide.[43][44] It was given the brand name Ozempic. Clinical trials started in January 2016 and ended in May 2017.[19][45]
In March 2021, in a phase III randomized, double-blind trial, 1,961 adults with a body mass index of 30 or greater were assigned in a 2:1 ratio to a treatment with once-weekly subcutaneous semaglutide or placebo, plus lifestyle intervention. The trials occurred at 129 sites in 16 countries in Asia, Europe, North America, and South America. The mean percentage change in body weight at week 68 was −14.9% in the semaglutide group vs −2.4% with placebo, for an estimated treatment difference of −12.4 percentage points (95% CI, −13.4 to −11.5).[46][47][48][49]
A 2022 review of anti-obesity treatments found that semaglutide as well as tirzepatide (which has an overlapping mechanism of action) were more promising than previous anti-obesity drugs, although less effective than bariatric surgery.[50]
In March 2023, a Novo Nordisk official said that patients using semaglutide to lose weight can regain their original weight within 5 years of stopping treatment.[51]
The US Food and Drug Administration (FDA) approved semaglutide based on evidence from seven clinical trials of 4087 participants with type 2 diabetes.[25] The trials were conducted at 536 sites in 33 countries, including Canada, Mexico, the Russian Federation, Ukraine, Turkey, India, South Africa, Japan, Hong Kong, multiple European countries, Argentina, and the United States.[25] In two of these trials (NCT #02054897 and NCT#02305381), participants were randomly assigned to receive either semaglutide or placebo injection weekly.[25] Neither the participant nor the health care provider knew which treatment was being given until after the trials were completed.[25] Treatment was given for 30 weeks.[25] In the other five trials (NCT #01930188, 01885208, 02128932, 02207374, 02254291), participants were randomly assigned to receive either semaglutide or another antidiabetic medication, and the participant and provider knew which medication was being given in four trials.[25] Treatment was given for 30 weeks or 56 weeks.[25]
In each trial, HbA1c was measured from the start of the trial to the end of the trial and compared between the semaglutide group and the other groups.[25]
The FDA also considered data from one separate trial (NCT #01720446) of 3297 participants with type 2 diabetes who were at high risk for cardiovascular events.[25] This trial was conducted in 20 countries in Europe, the Russian Federation, Turkey, Brazil, Israel, Malaysia, Brazil, Mexico, Thailand, Taiwan, Canada, and the United States.[25] The participants were randomly assigned to receive semaglutide or placebo.[25] Neither the participant nor the health care provider knew which treatment was being given.[25] Treatment was given for 104 weeks (2 years), and the occurrence of cardiovascular events, including heart attacks, strokes and hospitalization due to unstable angina (near heart attack) were recorded and compared in the two groups of participants.[25]
In March 2024, the FDA expanded the indication for semaglutide (Wegovy) to reduce the risk of cardiovascular death, heart attack and stroke in adults with cardiovascular disease and either obesity or overweight.[30] The efficacy and safety for this new indication were studied in a multi-national, multi-center, placebo-controlled double-blind trial that randomly assigned over 17,600 participants to receive either semaglutide (Wegovy) or placebo.[30] Participants in both groups also received standard-of-care medical treatment (e.g., management of blood pressure and cholesterol) and healthy lifestyle counseling (including diet and physical activity).[30] Semaglutide (Wegovy) significantly reduced the risk of major adverse cardiovascular events (cardiovascular death, heart attack and stroke), which occurred in 6.5% of participants who received semaglutide (Wegovy) compared to 8% of participants who received placebo.[30]
Society and culture
[edit]Legal status
[edit]In December 2016, the US FDA New Drug Application (NDA) was filed, and in October 2017, the FDA Advisory Committee approved it unanimously.[52]
In December 2017, the injectable version with the brand name Ozempic was approved for use by people with diabetes in the United States,[26][53] and, in January 2018, in Canada.[54]
In February 2018, authorization was granted in the European Union,[16][55] in March 2018 in Japan,[56] and in August 2019 in Australia.[1][4]
In September 2019, a version that can be taken orally (Rybelsus) was approved for medical use in the United States,[57][58] and in the European Union in April 2020.[17]
In June 2021, a higher-dose version for injectable use sold under the brand name Wegovy was approved by the US Food and Drug Administration (FDA) as an anti-obesity medication for long-term weight management in adults.[15] In November 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) recommended to grant a marketing authorization for Wegovy[59] to Novo Nordisk A/S.[59] In January 2022, Wegovy was approved for medical use in the European Union.[18][60]
In January 2023, the label for Rybelsus was updated to reflect that it can be used as a first-line treatment for adults with type 2 diabetes.[61]
Grey market sellers offer unauthorized products claimed to be semaglutide online. This practice is illegal in the United States, but some buyers turn to unauthorized retailers due to being denied insurance coverage and not being able to afford the name brand drug.[62][63][64]
In October 2023, Belgium announced it was considering a temporary ban on Ozempic for use as a weight loss medication amid a sharp increase in demand leading to a shortage of the drug, which was expected to last into mid-2024. The government had previously advised medical professionals to prescribe the drug only to diabetics. A similar order had been issued by the government of the United Kingdom earlier that month, additionally prohibiting new prescriptions for type 2 diabetics.[65]
Generics
[edit]Semaglutide is expected to become patent-free in the United States no earlier than December 2031.[66]
The Chinese patent was scheduled to expire in 2026, but a court ruled in 2022 that all patents on semaglutide were invalid. Novo Nordisk appealed the ruling.[67]
In Brazil, the Supreme Court refused to extend semaglutide's patent protection, which expires in 2026.[68]
Economics
[edit]"Ozempic, the semaglutide injection used for T2D treatment, has a list price of $936 in the United States and $169 in Japan. Prices were $147 in Canada, $144 in Switzerland, $103 in Germany and Netherlands, $96 in Sweden, $93 in the United Kingdom, and $87 in Australia. France had the lowest price at $83." (21 August 2023; figures refer to a one-month supply)[69][70]
In the US, Wegovy has a list price of $1,349.02 per month as of 2022 suggesting that because of the high costs many people "who could most benefit from weight loss may be unable to afford such expensive drugs".[71] High costs of Ozempic prompted some insurance companies to investigate and refuse to cover patients with what the companies considered was insufficient evidence to support a diabetes diagnosis, alleging off-label prescribing for weight loss.[72]
In the UK, semaglutide is available on NHS prescription for diabetes at nominal or no cost to patients.[73] It is also available for obesity, limited to treatment for two years.[74]
High demand caused worldwide supply shortages of semaglutide in 2023;[72] new UK prescriptions were not issued during the shortage.
Economic impact on Danish society
By 2023, Novo Nordisk had become the most valuable corporation in the European Union, worth more than US$500 billion, and accounted for almost all recent economic growth in Denmark.[75]
Profits from Novo Nordisk generate returns for the Novo Nordisk Foundation, which holds the controlling stake in Novo Nordisk. The profits results in increased Danish tax revenues and employment. Novo Nordisk added 3,500 jobs in Denmark in 2022, bringing the total in the country to 21,000 employees, out of 59,000 worldwide.[76]
Counterfeits
[edit]In October 2023, there were reports of counterfeit Ozempic pens being sold in Europe.[77] The pens possibly contained insulin, and led to several people being hospitalised with hypoglycaemia and seizures.[78][79][80] In December 2023, the FDA issued a warning about counterfeit Ozempic in the United States.[81]
Compounded versions
[edit]Some compounded versions have been found to contain salts of semaglutide including the sodium and the acetate in an attempt to avoid the patent of the base semaglutide product. These are not evaluated for safety and effectiveness by and thus are considered not shown to be safe or effective by the US Food and Drug Administration (FDA).[82]
Research
[edit]A 2014 meta-analysis found that semaglutide may be effective in lowering liver enzymes (transaminitis) and improving certain radiologically observed features of metabolic dysfunction–associated steatotic liver disease.[83] French national health care insurance system database had previously suggested 1–3 years use of glucagon like peptide-1 receptor agonists like exenatide, liraglutide and dulaglutide may be linked with increased occurrence of thyroid cancer. Semaglutide belongs to the same family of medicine. A meta-analysis involving data from 37 randomized controlled trials and 19 real world studies (46,719 patients) showed that semaglutide use over 18 months was not associated with increased risks of any cancer, supported by a high grade of evidence.[84]
In July 2023, the Icelandic Medicines Agency reported two cases of suicidal thoughts and one case of self-injury of users of the injection, prompting a safety assessment of Ozempic,[85] Wegovy, Saxenda, and similar drugs.[86] In January 2024, a preliminary review conducted by the FDA confirmed no evidence had been found to suggest that the medicine causes suicidal thoughts or actions.[87][88]
Semaglutide has shown potential to reduce interest in alcohol consumption among users. Scientists speculate that semaglutide may influence brain regions involved in addiction and appetite regulation, although the exact mechanisms remain under study. Animal research has indicated that drugs similar to semaglutide can reduce alcohol intake.[89]
Eating disorders
[edit]Semaglutide and similar drugs, such as dulaglutide and liraglutide, have been used to treat binge eating disorder (BED), as they can successfully minimize obsessive thoughts about food and binging urges.[90][91] Some users of these drugs have reported significant reduction in what is colloquially known as "food noise" (constant, unstoppable thoughts about eating despite not being physically hungry), which can be a factor of BED.[92][93]
References
[edit]- ^ a b c "AusPAR: Semaglutide". Therapeutic Goods Administration (TGA). 2 December 2020. Archived from the original on 24 February 2022. Retrieved 23 February 2022.
- ^ a b "Rybelsus APMDS". Therapeutic Goods Administration (TGA). 22 February 2022. Retrieved 23 February 2022.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b "Summary for ARTG Entry:315107 Ozempic 1 mg semaglutide (rys) 1.34 mg/mL solution for injection pre-filled pen". Therapeutic Goods Administration (TGA). Archived from the original on 24 February 2022. Retrieved 6 June 2021.
- ^ "Summary for ARTG Entry: 346198 Rybelsus semaglutide 3 mg tablet blister pack". Therapeutic Goods Administration (TGA). Archived from the original on 24 February 2022. Retrieved 23 February 2022.
- ^ "Wegovy (Novo Nordisk Pharmaceuticals Pty Ltd)". Therapeutic Goods Administration (TGA). 7 October 2022. Archived from the original on 14 April 2023. Retrieved 7 April 2023.
- ^ Product Monograph Including Patient Medication Information – Ozempic semaglutide injection (PDF) (Report). Novo-Nordisk Canada. 21 August 2020 [Initial approval 4 January 2018]. Archived (PDF) from the original on 7 June 2021. Retrieved 6 June 2021.
- ^ Product Monograph Including Patient Medication Information – Rybelsus semaglutide tablets (PDF) (Report). Novo-Nordisk Canada. 30 March 2020. Archived (PDF) from the original on 14 December 2021. Retrieved 6 June 2021.
- ^ "Regulatory Decision Summary – Rybelsus". Health Canada. 23 October 2014. Archived from the original on 5 June 2022. Retrieved 4 June 2022.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ "Ozempic 0.25 mg solution for injection in pre-filled pen – Summary of Product Characteristics (SmPC)". (emc). 9 April 2021. Archived from the original on 6 June 2021. Retrieved 6 June 2021.
- ^ "Rybelsus – Summary of Product Characteristics (SmPC)". (emc). 25 November 2020. Archived from the original on 6 June 2021. Retrieved 6 June 2021.
- ^ a b c d e f g h i j k "Ozempic- semaglutide injection, solution". DailyMed. Archived from the original on 5 June 2021. Retrieved 5 June 2021.
- ^ a b c d e f g h i "Rybelsus- oral semaglutide tablet". DailyMed. Archived from the original on 5 June 2021. Retrieved 5 June 2021.
- ^ a b c d e f g h i j "Wegovy- semaglutide injection, solution". DailyMed. 4 June 2021. Archived from the original on 14 December 2021. Retrieved 11 March 2022.
- ^ a b c "Ozempic EPAR". European Medicines Agency (EMA). 8 February 2018. Archived from the original on 25 October 2020. Retrieved 26 September 2020.
- ^ a b c "Rybelsus EPAR". European Medicines Agency (EMA). 29 January 2020. Archived from the original on 14 August 2020. Retrieved 26 September 2020.
- ^ a b c d "Wegovy EPAR". European Medicines Agency. 6 January 2022. Archived from the original on 2 July 2022. Retrieved 11 March 2022.
- ^ a b Singh G, Krauthamer M, Bjalme-Evans M (January 2022). "Wegovy (semaglutide): a new weight loss drug for chronic weight management". Journal of Investigative Medicine. 70 (1): 5–13. doi:10.1136/jim-2021-001952. PMC 8717485. PMID 34706925.
- ^ Phillips A, Clements JN (February 2022). "Clinical review of subcutaneous semaglutide for obesity". Journal of Clinical Pharmacy and Therapeutics. 47 (2): 184–193. doi:10.1111/jcpt.13574. PMID 34964141. S2CID 245538758.
- ^ Amaro A, Sugimoto D, Wharton S (January 2022). "Efficacy and safety of semaglutide for weight management: evidence from the STEP program". Postgraduate Medicine. 134 (sup1): 5–17. doi:10.1080/00325481.2022.2147326. PMID 36691309. S2CID 256192982.
- ^ a b Doggrell SA (March 2018). "Semaglutide in type 2 diabetes - is it the best glucagon-like peptide 1 receptor agonist (GLP-1R agonist)?" (PDF). Expert Opinion on Drug Metabolism & Toxicology. 14 (3): 371–377. doi:10.1080/17425255.2018.1441286. PMID 29439603. S2CID 3421553. Archived (PDF) from the original on 5 May 2020. Retrieved 12 December 2019.
- ^ a b c Goldenberg RM, Steen O (March 2019). "Semaglutide: Review and Place in Therapy for Adults With Type 2 Diabetes". Canadian Journal of Diabetes. 43 (2): 136–145. doi:10.1016/j.jcjd.2018.05.008. PMID 30195966.
- ^ Hughes S, Neumiller JJ (January 2020). "Oral Semaglutide". Clinical Diabetes. 38 (1): 109–111. doi:10.2337/cd19-0079. PMC 6969659. PMID 31975761.
- ^ a b c d e f g h i j k l m n o p "Ozempic Drug Trial Snapshot". U.S. Food and Drug Administration (FDA). 5 December 2017. Archived from the original on 31 December 2023. Retrieved 31 December 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "Drug Approval Package: Ozempic (semaglutide) Injection". U.S. Food and Drug Administration (FDA). 16 January 2018. Archived from the original on 1 March 2021. Retrieved 26 September 2020.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Semaglutide - Drug Usage Statistics". ClinCalc. Archived from the original on 8 October 2022. Retrieved 14 January 2024.
- ^ "FDA Approves New Drug Treatment for Chronic Weight Management, First Since 2014". U.S. Food and Drug Administration (FDA) (Press release). 4 June 2021. Archived from the original on 4 June 2021. Retrieved 5 June 2021.
- ^ a b c d e "FDA Approves First Treatment to Reduce Risk of Serious Heart Problems Specifically in Adults with Obesity or Overweight". U.S. Food and Drug Administration (FDA) (Press release). 8 March 2024. Retrieved 12 March 2024.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Sodhi M, Rezaeianzadeh R, Kezouh A, Etminan M (October 2023). "Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss". The Journal of the American Medical Association. 330 (18): 1795–1797. doi:10.1001/jama.2023.19574. PMC 10557026. PMID 37796527.
- ^ Sande CM, Tondi Resta I, Livolsi VA (March 2023). "The Thyroid Pathologist Meets Therapeutic Pharmacology". Endocrine Pathology. 34 (1): 48–56. doi:10.1007/s12022-023-09749-1. PMC 9890439. PMID 36723855.
- ^ Bjerre Knudsen L, Madsen LW, Andersen S, Almholt K, de Boer AS, Drucker DJ, et al. (April 2010). "Glucagon-like Peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation". Endocrinology. 151 (4): 1473–1486. doi:10.1210/en.2009-1272. PMID 20203154. S2CID 20934882.
- ^ Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. (November 2016). "Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes". The New England Journal of Medicine. 375 (19): 1834–1844. doi:10.1056/NEJMoa1607141. PMID 27633186.
- ^ Li Y, Hansotia T, Yusta B, Ris F, Halban P, Drucker D (2003). "Glucagon-like Peptide-1 Receptor Signaling Modulates β Cell Apoptosis". The Journal of Biological Chemistry. 278 (1): 471–478. doi:10.1074/jbc.M209423200. PMID 12409292.
- ^ Dhillon S (February 2018). "Semaglutide: First Global Approval". Drugs. 78 (2): 275–284. doi:10.1007/s40265-018-0871-0. PMID 29363040. S2CID 46851453.
- ^ Blundell J, Finlayson G, Axelsen M, Flint A, Gibbons C, Kvist T, et al. (September 2017). "Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity". Diabetes, Obesity & Metabolism. 19 (9): 1242–1251. doi:10.1111/dom.12932. PMC 5573908. PMID 28266779.
- ^ a b c Lau J, Bloch P, Schäffer L, Pettersson I, Spetzler J, Kofoed J, et al. (September 2015). "Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide". Journal of Medicinal Chemistry. 58 (18): 7370–80. doi:10.1021/acs.jmedchem.5b00726. PMID 26308095. S2CID 20228358.
- ^ a b c Gotfredsen CF, Mølck AM, Thorup I, Nyborg NC, Salanti Z, Knudsen LB, et al. (July 2014). "The human GLP-1 analogs liraglutide and semaglutide: absence of histopathological effects on the pancreas in nonhuman primates". Diabetes. 63 (7): 2486–97. doi:10.2337/db13-1087. PMID 24608440. S2CID 35102048.
- ^ Kapitza C, Nosek L, Jensen L, Hartvig H, Jensen CB, Flint A (May 2015). "Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive, ethinylestradiol/levonorgestrel". Journal of Clinical Pharmacology. 55 (5): 497–504. doi:10.1002/jcph.443. PMC 4418331. PMID 25475122.
- ^ a b c Reynolds M. "What the Scientists Who Pioneered Weight-Loss Drugs Want You to Know". Wired. ISSN 1059-1028. Retrieved 15 May 2024.
- ^ Müller MU, Preker A (4 January 2024). "Interview with Weight Loss Drugs Inventor Lotte Bjerre Knudsen: "I'm Experimenting with Periodic Fasting"". Der Spiegel. ISSN 2195-1349. Retrieved 15 May 2024.
- ^ Clinical trial number NCT00696657 for "A Randomised Controlled Clinical Trial in Type 2 Diabetes Comparing Semaglutide to Placebo and Liraglutide" at ClinicalTrials.gov
- ^ Kalra S, Gupta Y (July 2015). "Once-weekly glucagon-like peptide 1 receptor agonists". The Journal of the Pakistan Medical Association. 65 (7): 796–8. PMID 26160096. Archived from the original on 30 September 2022. Retrieved 10 April 2022.
- ^ "Efficacy and Safety of Semaglutide Versus Dulaglutide as add-on to Metformin in Subjects With Type 2 Diabetes". ClinicalTrials.gov. October 2019. Archived from the original on 31 July 2018. Retrieved 3 March 2023.
- ^ Wilding JP, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al. (March 2021). "Once-Weekly Semaglutide in Adults with Overweight or Obesity". The New England Journal of Medicine. 384 (11): 989–1002. doi:10.1056/NEJMoa2032183. PMID 33567185. S2CID 231883214.
- ^ Blum D (22 November 2022). "What Is Ozempic and Why Is It Getting So Much Attention?". The New York Times. ISSN 0362-4331. Archived from the original on 29 March 2023. Retrieved 30 March 2023.
- ^ Garvey WT, Batterham RL, Bhatta M, Buscemi S, Christensen LN, Frias JP, et al. (October 2022). "Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial". Nature Medicine. 28 (10): 2083–2091. doi:10.1038/s41591-022-02026-4. PMC 9556320. PMID 36216945.
- ^ Dillinger K (29 March 2023). "WHO advisers to consider whether obesity medication should be added to Essential Medicines List". CNN. Archived from the original on 30 March 2023. Retrieved 30 March 2023.
- ^ Müller TD, Blüher M, Tschöp MH, DiMarchi RD (March 2022). "Anti-obesity drug discovery: advances and challenges". Nature Reviews. Drug Discovery. 21 (3): 201–223. doi:10.1038/s41573-021-00337-8. PMC 8609996. PMID 34815532.
- ^ "Novo Nordisk says stopping obesity drug may cause full weight regain in 5 years". Reuters. 30 March 2023. Archived from the original on 10 June 2023. Retrieved 12 February 2024.
- ^ "Development Status and FDA Approval Process for semaglutide". Drugs.com. 2017. Archived from the original on 24 October 2017. Retrieved 24 October 2017.
- ^ "Ozempic (semaglutide) approved in the US" (Press release). Novo Nordisk. 5 December 2017. Archived from the original on 5 June 2021. Retrieved 5 June 2021.
- ^ "Regulatory Decision Summary – Ozempic". Health Canada. 23 October 2014. Archived from the original on 17 May 2019. Retrieved 2 April 2019.
- ^ "Novo Nordisk A/S: Ozempic (semaglutide) approved in the EU for the treatment of type 2 diabetes" (Press release). Novo Nordisk A/S. 9 February 2018. Archived from the original on 2 April 2019. Retrieved 19 August 2018 – via GlobeNewswire.
- ^ "Ozempic approved in Japan for the treatment of type 2 diabetes" (Press release). Novo Nordisk A/S. 23 March 2018. Archived from the original on 2 April 2019. Retrieved 2 April 2019 – via GlobeNewswire.
- ^ "Drug Approval Package: Rybelsus". U.S. Food and Drug Administration (FDA). 10 June 2020. Archived from the original on 2 November 2020. Retrieved 26 September 2020.
- ^ "FDA approves first oral GLP-1 treatment for type 2 diabetes" (Press release). U.S. Food and Drug Administration (FDA). 20 September 2019. Archived from the original on 23 September 2019. Retrieved 20 September 2019.
- ^ a b "Wegovy : Pending EC decision". European Medicines Agency. 11 November 2021. Archived from the original on 13 November 2021. Retrieved 13 November 2021. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Wegovy Product information". Union Register of medicinal products. Archived from the original on 30 October 2022. Retrieved 3 March 2023.
- ^ "Novo Nordisk announces FDA approval of label update for Rybelsus (semaglutide) allowing use as a first-line option for adults with type 2 diabetes" (Press release). Novo Nordisk. 12 January 2023. Archived from the original on 16 January 2023. Retrieved 16 January 2023 – via PR Newswire.
- ^ Lovelace Jr B (3 May 2023). "Safety worries over copycat versions of Ozempic and Wegovy prompt state crackdowns". NBC News. Archived from the original on 25 September 2023. Retrieved 26 September 2023.
- ^ Gilbert D (19 September 2023). "Inside the gold rush to sell cheaper imitations of Ozempic". Washington Post. Retrieved 26 September 2023.
- ^ Lovelace Jr B, Lewis R, Kopf M (19 March 2023). "The high price of Ozempic is pushing many to unregulated, copycat drugs for weight loss". NBC News. Archived from the original on 26 September 2023. Retrieved 26 September 2023.
- ^ Strauss M (24 October 2023). "Belgium plans temporary ban on use of Ozempic for weight loss". Reuters. Archived from the original on 24 October 2023. Retrieved 24 October 2023.
- ^ "OZEMPIC Drug Patent Profile". Drug Patent Watch. Retrieved 23 March 2024.
- ^ He L (7 June 2023). "Ozempic is taking China by storm. Drugmakers are scrambling to boost supplies". CNN Business. Archived from the original on 15 December 2023. Retrieved 1 December 2023.
- ^ Masters W (9 May 2023). "Brazilian federal court denies semaglutide patent extension". Pharmaceutical Technology. Archived from the original on 6 January 2024. Retrieved 1 December 2023.
- ^ "U.S. Leads the world in list prices for diabetes, antiobesity meds". 21 August 2023. Archived from the original on 20 January 2024. Retrieved 20 January 2024.
- ^ "Charted: The cost of weight-loss drugs in the US vs. Other countries". Archived from the original on 20 January 2024. Retrieved 8 April 2024.
- ^ Kolata G (28 April 2022). "Patients Taking Experimental Obesity Drug Lost More Than 50 Pounds, Maker Claims". The New York Times. Archived from the original on 27 June 2022. Retrieved 13 May 2022.
- ^ a b Gilbert D (12 June 2023). "Insurers clamping down on doctors who prescribe Ozempic for weight loss". The Washington Post.
- ^ "NICE recommended weight-loss drug to be made available in specialist NHS services". National Institute for Health and Care Excellence (NICE). 8 March 2023. Archived from the original on 9 March 2023.
- ^ Marcus R (6 June 2023). "I lost 40 pounds on Ozempic. But I'm left with even more questions". The Washington Post. Archived from the original on 7 June 2023.
- ^ Verrender I (19 December 2023). "How Ozempic could affect the health of the global economy in more ways than one". ABC News. Archived from the original on 18 December 2023. Retrieved 18 December 2023.
- ^ "Novo Nordisk's Wegovy bonanza looms large in Denmark". Reuters. Retrieved 5 April 2024.
- ^ "EMA alerts EU patients and healthcare professionals to reports of falsified Ozempic pens". European Medicines Agency (Press release). 18 October 2023. Archived from the original on 4 January 2024. Retrieved 6 January 2024.
- ^ "Ozempic (semaglutide) and Saxenda (liraglutide): vigilance required due to potentially harmful falsified products". Medicines and Healthcare products Regulatory Agency. 23 November 2023. Archived from the original on 26 December 2023. Retrieved 6 January 2024.
- ^ "European regulator warns EU, UK about fake Ozempic pens". Reuters. 18 October 2023. Archived from the original on 24 October 2023. Retrieved 24 October 2023.
- ^ "Several hospitalised in Austria after using suspected fake diabetes drug". The Guardian. Reuters. 24 October 2023. Archived from the original on 24 October 2023. Retrieved 24 October 2023.
- ^ "FDA warns consumers not to use counterfeit Ozempic (semaglutide) found in U.S. drug supply chain". U.S. Food and Drug Administration (FDA). 21 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023.
- ^ "Medications Containing Semaglutide Marketed for Type 2 Diabetes or Weight Loss". U.S. Food and Drug Administration (FDA). 21 December 2023. Archived from the original on 29 December 2023. Retrieved 29 December 2023.
- ^ Dutta D, Kumar M, Shivaprasad KS, Kumar A, Sharma M (June 2022). "Impact of semaglutide on biochemical and radiologic measures of metabolic-dysfunction associated fatty liver disease across the spectrum of glycaemia: A meta-analysis". Diabetes & Metabolic Syndrome. 16 (6): 102539. doi:10.1016/j.dsx.2022.102539. PMID 35709586. S2CID 249584781.
- ^ Nagendra L, Bg H, Sharma M, Dutta D (July 2023). "Semaglutide and cancer: A systematic review and meta-analysis.". Diabetes Metab Syndr. 17 (9): 102834. doi:10.1016/j.dsx.2023.102834. PMID 37531876. S2CID 260261877.
- ^ Blanchet B (27 July 2023). "Ozempic, Drugs for Weight Loss Being Reviewed for Links to Suicide Risk". People. Archived from the original on 28 July 2023. Retrieved 28 July 2023.
- ^ "Weight-loss jabs investigated for suicide risk". BBC News. 10 July 2023. Archived from the original on 10 July 2023. Retrieved 10 July 2023.
- ^ "FDA reports no link between weight-loss drugs and suicidal thoughts". The Washington Post. Archived from the original on 14 January 2024. Retrieved 19 January 2024.
- ^ "Update on FDA's ongoing evaluation of reports of suicidal thoughts or actions in patients taking a certain type of medicines approved for type 2 diabetes and obesity". U.S. Food and Drug Administration (FDA). 11 January 2024. Archived from the original on 19 January 2024. Retrieved 19 January 2024.
- ^ Blum D (24 February 2023). "Some People on Ozempic Lose the Desire to Drink. Scientists Are Asking Why". The New York Times. Retrieved 20 July 2024.
- ^ Hayashi D, Edwards C, Emond JA, Gilbert-Diamond D, Butt M, Rigby A, et al. (November 2023). Trakada G (ed.). "What Is Food Noise? A Conceptual Model of Food Cue Reactivity". Nutrients. 15 (22): 4809. doi:10.3390/nu15224809. PMC 10674813. PMID 38004203.
- ^ Järvinen A, Laine MK, Tikkanen R, Castrén ML (2019). "Beneficial Effects of GLP-1 Agonist in a Male With Compulsive Food-Related Behavior Associated With Autism". Frontiers in Psychiatry. 10: 97. doi:10.3389/fpsyt.2019.00097. PMC 6405420. PMID 30881319.
- ^ Kuhn C (25 September 2023). "Patients say drugs like Ozempic help with 'food noise.' Here's what that means". PBS. Archived from the original on 13 January 2024. Retrieved 13 January 2024.
- ^ Blum D (21 June 2023). "People on Drugs Like Ozempic Say Their 'Food Noise' Has Disappeared". The New York Times. Archived from the original on 11 January 2024. Retrieved 13 January 2024.
External links
[edit]- "Semaglutide tablet". MedlinePlus.
- Clinical trial number NCT03574597 for "Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT)" at ClinicalTrials.gov