| |
| Names | |
|---|---|
| Other names
Manganese pentafluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| |
| |
| Properties | |
| MnF5 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
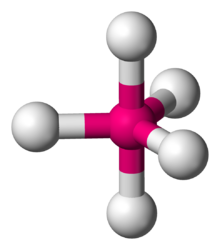
Manganese pentafluoride is a binary hypothetical inorganic compound of manganese and fluorine with a chemical formula of MnF5.[1][2]
Manganese(V) fluoride is believed to be unstable both in a condensed state and gaseous phase.[3] Computational studies predict it will have trigonal bipyramidal molecular geometry.[2]