| |
| Names | |
|---|---|
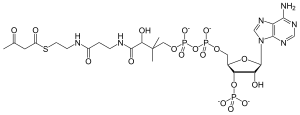
| IUPAC name
3′-O-Phosphonoadenosine 5′-[(3R)-3-hydroxy-2,2-dimethyl-4-oxo-4-{[3-oxo-3-({2-[(3-oxobutanoyl)sulfanyl]ethyl}amino)propyl]amino}butyl dihydrogen diphosphate]
| |
| Systematic IUPAC name
O1-{[(2R,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)oxolan-2-yl]methyl} O3-[(3R)-3-hydroxy-2,2-dimethyl-4-oxo-4-{[3-oxo-3-({2-[(3-oxobutanoyl)sulfanyl]ethyl}amino)propyl]amino}butyl] dihydrogen diphosphate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.014.378 |
| MeSH | acetoacetyl+CoA |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C25H40N7O18P3S | |
| Molar mass | 851.61 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Acetoacetyl CoA is the precursor of HMG-CoA in the mevalonate pathway, which is essential for cholesterol biosynthesis. It also takes a similar role in the ketone bodies synthesis (ketogenesis) pathway of the liver.[1] In the ketone bodies digestion pathway (in the tissue), it is no longer associated with having HMG-CoA as a product or as a reactant.
It is created from acetyl-CoA, a thioester, which reacts with the enolate of a second molecule of acetyl-CoA in a Claisen condensation reaction,[2] and it is acted upon by HMG-CoA synthase to form HMG-CoA.[1] During the metabolism of leucine, this last reaction is reversed. Some individuals may experience Acetoacetyl-CoA deficiency.[3] This deficiency is classified as a disorder ketone body and isoleucine metabolism that can be inherited.[citation needed] Additional mutations include those with the enzymes within pathways related to Acetoacetyl CoA, including Beta-Ketothiolase deficiency and Mitochondrial 3-hydroxy-3-methylglutaryl-CoA Synthase mutation.

Additionally, it reacts with NADPH-dependent acetoacetyl-coenzyme A reductase, also known as PhaB, in a pathway that produces polyester polyhydroxyalkanoate (PHA). The reduction of acetoacetyl-coA by Pha creates (R)-3-hydroxybutyryl-CoA, which polymerizes to PHA.[4] The pathway is present in bacteria such as Ralstonia eutropha and the PCC6803 strain of Synechocystis.[5] Mover over, Acetoacetyl-CoA is involved with neuronal development involving lipogenesis and providing fats and cholesterol for neuronal cells.