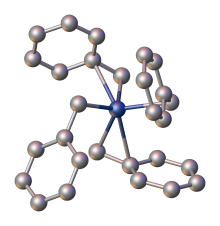
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure R−CH2−C6H5. Benzyl features a benzene ring (C6H6) attached to a methylene group (−CH2−) group.[1]

In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure R−CH2−C6H5. Benzyl features a benzene ring (C6H6) attached to a methylene group (−CH2−) group.[1]
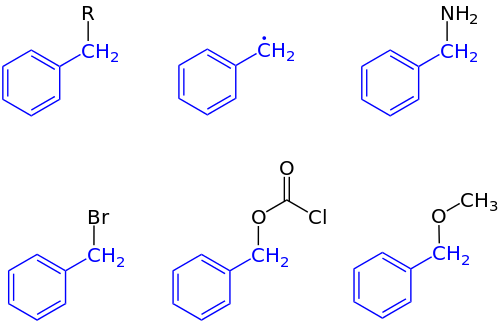
In IUPAC nomenclature, the prefix benzyl refers to a C6H5CH2 substituent, for example benzyl chloride or benzyl benzoate. Benzyl is not to be confused with phenyl with the formula C6H5. The term benzylic is used to describe the position of the first carbon bonded to a benzene or other aromatic ring. For example, (C6H5)(CH3)2C+ is referred to as a "benzylic" carbocation. The benzyl free radical has the formula C6H5CH2•. The benzyl cation or phenylcarbenium ion is the carbocation with formula C6H5CH+2; the benzyl anion or phenylmethanide ion is the carbanion with the formula C6H5CH−2. None of these species can be formed in significant amounts in the solution phase under normal conditions, but they are useful referents for discussion of reaction mechanisms and may exist as reactive intermediates.
Benzyl is most commonly abbreviated Bn. For example, benzyl alcohol can be represented as BnOH. Less common abbreviations are Bzl and Bz, the latter of which is ambiguous as it is also the standard abbreviation for the benzoyl group C6H5C(O)−. Likewise, benzyl should not be confused with the phenyl group C6H5−, abbreviated Ph.
The enhanced reactivity of benzylic positions is attributed to the low bond dissociation energy for benzylic C−H bonds. Specifically, the bond C6H5CH2−H is about 10–15% weaker than other kinds of C−H bonds. The neighboring aromatic ring stabilizes benzyl radicals. The data tabulated below compare benzylic C−H bond to related C−H bond strengths.
| Bond | Bond | Bond-dissociation energy[2][3] | Comment | |
|---|---|---|---|---|
| (kcal/mol) | (kJ/mol) | |||
| C6H5CH2−H | benzylic C−H bond | 90 | 377 | akin to allylic C−H bonds such bonds show enhanced reactivity |
| H3C−H | methyl C−H bond | 105 | 439 | one of the strongest aliphatic C−H bonds |
| C2H5−H | ethyl C−H bond | 101 | 423 | slightly weaker than H3C−H |
| C6H5−H | phenyl C−H bond | 113 | 473 | comparable to vinyl radical, rare |
| CH2=CHCH2−H | allylic C–H bond | 89 | 372 | similar to benzylic C-H |
| (C6H4)2CH−H | fluorenyl C–H bond | 80 | more activated vs diphenylmethyle (pKa = 22.6) | |
| (C6H5)2CH−H | diphenylmethyl C–H bond | 82 | "doubly benzylic" (pKa = 32.2) | |
| (C6H5)3C−H | trityl C–H bond | 81 | 339 | "triply benzylic" |
The weakness of the C−H bond reflects the stability of the benzylic radical. For related reasons, benzylic substituents exhibit enhanced reactivity, as in oxidation, free radical halogenation, or hydrogenolysis. As a practical example, in the presence of suitable catalysts, p-xylene oxidizes exclusively at the benzylic positions to give terephthalic acid:
Millions of tonnes of terephthalic acid are produced annually by this method.[4]
In a few cases, these benzylic transformations occur under conditions suitable for lab synthesis. The Wohl-Ziegler reaction will brominate a benzylic C–H bond: (ArCHR2 → ArCBrR2).[5] Any non-tertiary benzylic alkyl group will be oxidized to a carboxyl group by aqueous potassium permanganate (KMnO4) or concentrated nitric acid (HNO3): (ArCHR2 → ArCOOH).[6] Finally, the complex of chromium trioxide and 3,5-dimethylpyrazole (CrO3−dmpyz) will selectively oxidize a benzylic methylene group to a carbonyl: (ArCH2R → ArC(O)R).[7] 2-iodoxybenzoic acid in DMSO performs similarly.[8]
Benzyl groups are occasionally employed as protecting groups in organic synthesis. Their installation and especially their removal require relatively harsh conditions, so benzyl is not typically preferred for protection.[9]
Benzyl is commonly used in organic synthesis as a robust protecting group for alcohols and carboxylic acids.
Benzyl ethers can be removed under reductive conditions, oxidative conditions, and the use of Lewis acids.[9]
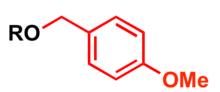
p-Methoxybenzyl (PMB) is used as a protecting group for alcohols in organic synthesis (4-Methoxybenzylthiol is used to protect thiols).
The benzyl group is occasionally used as a protecting group for amines in organic synthesis. Other methods exist.[9]