| Diphtheria | |
|---|---|
 | |
| An adherent, dense, grey pseudomembrane covering the tonsils is classically seen in diphtheria. | |
| Specialty | Infectious disease |
| Symptoms | Sore throat, fever, barking cough[1] |
| Complications | Myocarditis, Peripheral neuropathy, Proteinuria |
| Usual onset | 2–5 days post-exposure[2] |
| Causes | Corynebacterium diphtheriae (spread by direct contact and through the air)[2] |
| Diagnostic method | Examination of throat, culture[1] |
| Prevention | Diphtheria vaccine[2] |
| Treatment | Antibiotics, tracheostomy[2] |
| Prognosis | 5–10% risk of death |
| Frequency | 4,500 (reported 2015)[3] |
| Deaths | 2,100 (2015)[4] |
Diphtheria is an infection caused by the bacterium Corynebacterium diphtheriae.[2] Most infections are asymptomatic or have a mild clinical course, but in some outbreaks, the mortality rate approaches 10%.[1] Signs and symptoms may vary from mild to severe,[1] and usually start two to five days after exposure.[2] Symptoms often develop gradually, beginning with a sore throat and fever.[1] In severe cases, a grey or white patch develops in the throat,[2][1] which can block the airway, and create a barking cough similar to what is observed in croup.[1] The neck may also swell, in part due to the enlargement of the facial lymph nodes.[2] Diphtheria can also involve the skin, eyes, or genitals, and can cause[2][1] complications, including myocarditis (which in itself can result in an abnormal heart rate), inflammation of nerves (which can result in paralysis), kidney problems, and bleeding problems due to low levels of platelets.[2]
Diphtheria is usually spread between people by direct contact, through the air, or through contact with contaminated objects.[2][5] Asymptomatic transmission and chronic infection are also possible.[2] Different strains of C. diphtheriae are the main cause in the variability of lethality,[2] as the lethality and symptoms themselves are caused by the exotoxin produced by the bacteria.[1] Diagnosis can often be made based on the appearance of the throat with confirmation by microbiological culture.[1] Previous infection may not protect against reinfection.[1]
A diphtheria vaccine is effective for prevention, and is available in a number of formulations.[2] Three or four doses, given along with tetanus vaccine and pertussis vaccine, are recommended during childhood.[2] Further doses of the diphtheria–tetanus vaccine are recommended every ten years.[2] Protection can be verified by measuring the antitoxin level in the blood.[2] Diphtheria can be prevented in those exposed, as well as treated with the antibiotics erythromycin or benzylpenicillin.[2]In severe cases a tracheotomy is sometimes needed to open the airway.[1]
In 2015, 4,500 cases were officially reported worldwide, down from nearly 100,000 in 1980.[3] About a million cases a year are believed to have occurred before the 1980s.[1] Diphtheria currently occurs most often in sub-Saharan Africa, India, and Indonesia.[1][6] In 2015, it resulted in 2,100 deaths, down from 8,000 deaths in 1990.[4][7] In areas where it is still common, children are most affected.[1] It is rare in the developed world due to widespread vaccination, but can re-emerge if vaccination rates decrease.[1][8] In the United States, 57 cases were reported between 1980 and 2004.[2] Death occurs in 5–10% of those diagnosed.[2] The disease was first described in the 5th century BC by Hippocrates.[2] The bacterium was identified in 1882 by Edwin Klebs.[2]
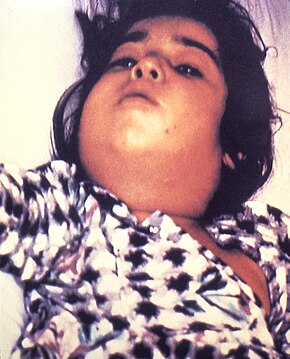

The symptoms of diphtheria usually begin two to seven days after infection. They include fever of 38 °C (100.4 °F) or above; chills; fatigue; bluish skin coloration (cyanosis); sore throat; hoarseness; cough; headache; difficulty swallowing; painful swallowing; difficulty breathing; rapid breathing; foul-smelling and bloodstained nasal discharge; and lymphadenopathy.[9][10] Within two to three days, diphtheria may destroy healthy tissues in the respiratory system. The dead tissue forms a thick, gray coating that can build up in the throat or nose. This thick gray coating is called a "pseudomembrane." It can cover tissues in the nose, tonsils, voice box, and throat, making it very hard to breathe and swallow.[11] Symptoms can also include cardiac arrhythmias, myocarditis, and cranial and peripheral nerve palsies.[12]
Laryngeal diphtheria can lead to a characteristic swollen neck and throat, or "bull neck." The swollen throat is often accompanied by a serious respiratory condition, characterized by a brassy or "barking" cough, stridor, hoarseness, and difficulty breathing; and historically referred to variously as "diphtheritic croup,"[13] "true croup,"[14][15] or sometimes simply as "croup."[16] Diphtheritic croup is extremely rare in countries where diphtheria vaccination is customary. As a result, the term "croup" nowadays most often refers to an unrelated viral illness that produces similar but milder respiratory symptoms.[17]
Human-to-human transmission of diphtheria typically occurs through the air when an infected individual coughs or sneezes. Breathing in particles released from the infected individual leads to infection.[18] Contact with any lesions on the skin can also lead to transmission of diphtheria, but this is uncommon.[19] Indirect infections can occur, as well. If an infected individual touches a surface or object, the bacteria can be left behind and remain viable. Also, some evidence indicates diphtheria has the potential to be zoonotic, but this has yet to be confirmed. Corynebacterium ulcerans has been found in some animals, which would suggest zoonotic potential.[20]
Diphtheria toxin (DT) is produced only by C. diphtheriae infected with a certain type of bacteriophage.[21][22] Toxinogenicity is determined by phage conversion (also called lysogenic conversion); i.e., the ability of the bacterium to make DT changes as a consequence of infection by a particular phage. DT is encoded by the tox gene. Strains of corynephage are either tox+ (e.g., corynephage β) or tox− (e.g., corynephage γ). The tox gene becomes integrated into the bacterial genome.[23] The chromosome of C. diphtheriae has two different but functionally equivalent bacterial attachment sites (attB) for integration of β prophage into the chromosome.
The diphtheria toxin precursor is a protein of molecular weight 60 kDa. Certain proteases, such as trypsin, selectively cleave DT to generate two peptide chains, amino-terminal fragment A (DT-A) and carboxyl-terminal fragment B (DT-B), which are held together by a disulfide bond.[23] DT-B is a recognition subunit that gains entry of DT into the host cell by binding to the EGF-like domain of heparin-binding EGF-like growth factor on the cell surface. This signals the cell to internalize the toxin within an endosome via receptor-mediated endocytosis. Inside the endosome, DT is split by a trypsin-like protease into DT-A and DT-B. The acidity of the endosome causes DT-B to create pores in the endosome membrane, thereby catalysing the release of DT-A into the cytoplasm.[23]
Fragment A inhibits the synthesis of new proteins in the affected cell by catalyzing ADP-ribosylation of elongation factor EF-2—a protein that is essential to the translation step of protein synthesis. This ADP-ribosylation involves the transfer of an ADP-ribose from NAD+ to a diphthamide (a modified histidine) residue within the EF-2 protein. Since EF-2 is needed for the moving of tRNA from the A-site to the P-site of the ribosome during protein translation, ADP-ribosylation of EF-2 prevents protein synthesis.[24]
ADP-ribosylation of EF-2 is reversed by giving high doses of nicotinamide (a form of vitamin B3), since this is one of the reaction's end products, and high amounts drive the reaction in the opposite direction.[25]
The current clinical case definition of diphtheria used by the United States' Centers for Disease Control and Prevention is based on both laboratory and clinical criteria.
Empirical treatment should generally be started in a patient in whom suspicion of diphtheria is high.
|
Main article: Diphtheria vaccine |
Vaccination against diphtheria is commonly done in infants, and delivered as a combination vaccine, such as a DPT vaccine (diphtheria, pertussis, tetanus). Pentavalent vaccines, which vaccinate against diphtheria and four other childhood diseases simultaneously, are frequently used in disease prevention programs in developing countries by organizations such as UNICEF.[26]
The disease may remain manageable, but in more severe cases, lymph nodes in the neck may swell, and breathing and swallowing are more difficult. People in this stage should seek immediate medical attention, as obstruction in the throat may require intubation or a tracheotomy. Abnormal cardiac rhythms can occur early in the course of the illness or weeks later, and can lead to heart failure. Diphtheria can also cause paralysis in the eye, neck, throat, or respiratory muscles. Patients with severe cases are put in a hospital intensive care unit, and given diphtheria antitoxin (consisting of antibodies isolated from the serum of horses that have been challenged with diphtheria toxin).[27] Since antitoxin does not neutralize toxin that is already bound to tissues, delaying its administration increases risk of death. Therefore, the decision to administer diphtheria antitoxin is based on clinical diagnosis, and should not await laboratory confirmation.[28]
Antibiotics have not been demonstrated to affect healing of local infection in diphtheria patients treated with antitoxin. Antibiotics are used in patients or carriers to eradicate C. diphtheriae, and prevent its transmission to others. The Centers for Disease Control and Prevention (CDC) recommends[29] either:
In cases that progress beyond a throat infection, diphtheria toxin spreads through the blood, and can lead to potentially life-threatening complications that affect other organs, such as the heart and kidneys. Damage to the heart caused by the toxin affects the heart's ability to pump blood, or the kidneys' ability to clear wastes. It can also cause nerve damage, eventually leading to paralysis. About 40–50% of those left untreated can die.[citation needed][30]


Diphtheria is fatal in 5–10% of cases. In children under five years and adults over 40 years, the fatality rate may be as much as 20%.[28] In 2013, it resulted in 3,300 deaths, down from 8,000 deaths in 1990.[7] Better standards of living, mass immunization, improved diagnosis, prompt treatment, and more effective health care have led to a decrease in cases worldwide.[31]
In 1613, Spain experienced an epidemic of diphtheria, referred to as El Año de los Garrotillos (The Year of Strangulations).[31]
In 1705, the Mariana Islands experienced an epidemic of diphtheria and typhus simultaneously, reducing the population to about 5,000 people.[32]
In 1735, a diphtheria epidemic swept through New England.[33]
Before 1826, diphtheria was known by different names across the world. In England, it was known as "Boulogne sore throat," as the illness had spread from France. In 1826, Pierre Bretonneau gave the disease the name diphthérite (from Greek διφθέρα, diphthera 'leather'), describing the appearance of pseudomembrane in the throat.[34][35]
In 1856, Victor Fourgeaud described an epidemic of diphtheria in California.[36]
In 1878, Princess Alice (Queen Victoria's second daughter) and her family became infected with diphtheria; Princess Alice and her four-year-old daughter, Princess Marie, both died.[37][self-published source]
In 1883, Edwin Klebs identified the bacterium causing diphtheria,[38] and named it Klebs–Loeffler bacterium. The club shape of this bacterium helped Edwin to differentiate it from other bacteria. Over time, it has been called Microsporon diphtheriticum, Bacillus diphtheriae, and Mycobacterium diphtheriae. Current nomenclature is Corynebacterium diphtheriae.[39]
In 1884, German bacteriologist Friedrich Loeffler became the first person to cultivate C. diphtheriae.[40] He used Koch's postulates to prove association between C. diphtheriae and diphtheria. He also showed that the bacillus produces an exotoxin.[citation needed]

In 1885, Joseph P. O'Dwyer introduced the O'Dwyer tube for laryngeal intubation in patients with an obstructed larynx. It soon replaced tracheostomy as the emergency diphtheric intubation method.[41]
In 1888, Emile Roux and Alexandre Yersin showed that a substance produced by C. diphtheriae caused symptoms of diphtheria in animals.[42][43]
In 1890, Shibasaburō Kitasato and Emil von Behring immunized guinea pigs with heat-treated diphtheria toxin.[44] They also immunized goats and horses in the same way, and showed that an "antitoxin" made from serum of immunized animals could cure the disease in non-immunized animals. Behring used this antitoxin (now known to consist of antibodies that neutralize the toxin produced by C. diphtheriae) for human trials in 1891, but they were unsuccessful. Successful treatment of human patients with horse-derived antitoxin began in 1894, after production and quantification of antitoxin had been optimized.[45][27] In 1901, Von Behring won the first Nobel Prize in medicine for his work on diphtheria.[46]
In 1895, H. K. Mulford Company of Philadelphia started production and testing of diphtheria antitoxin in the United States.[47] Park and Biggs described the method for producing serum from horses for use in diphtheria treatment.[citation needed]
In 1897, Paul Ehrlich developed a standardized unit of measure for diphtheria antitoxin. This was the first ever standardization of a biological product, and played an important role in future developmental work on sera and vaccines.[48]
In 1901, 10 of 11 inoculated St. Louis children died from contaminated diphtheria antitoxin. The horse from which the antitoxin was derived died of tetanus. This incident, coupled with a tetanus outbreak in Camden, New Jersey,[49] played an important part in initiating federal regulation of biologic products.[50]
On 7 January 1904, Ruth Cleveland died of diphtheria at the age of 12 years in Princeton, New Jersey. Ruth was the eldest daughter of former President Grover Cleveland and the former First Lady, Frances Folsom.[citation needed]
In 1905, Franklin Royer, from Philadelphia's Municipal Hospital, published a paper urging timely treatment for diphtheria and adequate doses of antitoxin.[51] In 1906, Clemens Pirquet and Béla Schick described serum sickness in children receiving large quantities of horse-derived antitoxin.[52]
Between 1910 and 1911, Béla Schick developed the Schick test to detect pre-existing immunity to diphtheria in an exposed person. Only those who had not been exposed to diphtheria were vaccinated. A massive, five-year campaign was coordinated by Dr. Schick. As a part of the campaign, 85 million pieces of literature were distributed by the Metropolitan Life Insurance Company, with an appeal to parents to "Save your child from diphtheria." A vaccine was developed in the next decade, and deaths began declining significantly in 1924.[53]

In 1919, in Dallas, Texas, 10 children were killed and 60 others made seriously ill by toxic antitoxin which had passed the tests of the New York State Health Department. The manufacturer of the antitoxin, the Mulford Company of Philadelphia, paid damages in every case.[54]
During the 1920s, an annual estimate of 100,000 to 200,000 diphtheria cases and 13,000 to 15,000 deaths occurred in the United States.[28] Children represented a large majority of these cases and fatalities. One of the most infamous outbreaks of diphtheria occurred in 1925, in Nome, Alaska; the "Great Race of Mercy" to deliver diphtheria antitoxin is now celebrated by the Iditarod Trail Sled Dog Race.[55]
In 1926, Alexander Thomas Glenny increased the effectiveness of diphtheria toxoid (a modified version of the toxin used for vaccination) by treating it with aluminum salts.[56] Vaccination with toxoid was not widely used until the early 1930s.[57] In 1939, Dr. Nora Wattie, who was the Principal Medical Officer (Maternity and Child Welfare) of Glasgow between 1934– 1964,[58] introduced immunisation clinics across Glasgow, and promoted mother and child health education, resulting in virtual eradication of the infection in the city.[59]
Widespread vaccination pushed cases in the United States down from 4.4 per 100,000 inhabitants in 1932 to 2.0 in 1937. In Nazi Germany, where authorities preferred treatment and isolation over vaccination (until about 1939–1941), cases rose over the same period from 6.1 to 9.6 per 100,000 inhabitants.[60]
Between June 1942 and February 1943, 714 cases of diphtheria were recorded at Sham Shui Po Barracks, resulting in 112 deaths because the Imperial Japanese Army did not release supplies of anti-diphtheria serum.[61]
In 1943, diphtheria outbreaks accompanied war and disruption in Europe. The 1 million cases in Europe resulted in 50,000 deaths.[citation needed]
During 1948 in Kyoto, 68 of 606 children died after diphtheria immunization due to improper manufacture of aluminum phosphate toxoid.[62]
In 1974, the World Health Organization included DPT vaccine in their Expanded Programme on Immunization for developing countries.[63][64]
In 1975, an outbreak of cutaneous diphtheria in Seattle, Washington, was reported.[65]
After the breakup of the former Soviet Union in 1991, vaccination rates in its constituent countries fell so low that an explosion of diphtheria cases occurred. In 1991, 2,000 cases of diphtheria occurred in the USSR. Between 1991 and 1998, as many as 200,000 cases were reported in the Commonwealth of Independent States, and resulted in 5,000 deaths.[31] In 1994, the Russian Federation had 39,703 diphtheria cases. By contrast, in 1990, only 1,211 cases were reported.[66]
In early May 2010, a case of diphtheria was diagnosed in Port-au-Prince, Haiti, after the devastating 2010 Haiti earthquake. The 15-year-old male patient died while workers searched for antitoxin.[67]
In 2013, three children died of diphtheria in Hyderabad, India.[68]
In early June 2015, a case of diphtheria was diagnosed at Vall d'Hebron University Hospital in Barcelona, Spain. The six-year-old child who died of the illness had not been previously vaccinated due to parental opposition to vaccination.[69] It was the first case of diphtheria in the country since 1986, as reported by the Spanish daily newspaper El Mundo,[70] or from 1998, as reported by the WHO.[71]
In March 2016, a three-year-old girl died of diphtheria in the University Hospital of Antwerp, Belgium.[72]
In June 2016, a three-year-old, five-year-old, and seven-year-old girl died of diphtheria in Kedah, Malacca, and Sabah, Malaysia.[73]
In January 2017, more than 300 cases were recorded in Venezuela.[74][75]
In 2017, outbreaks occurred in a Rohingya refugee camp in Bangladesh, and amongst children unvaccinated due to the Yemeni Civil War.[76]
In November and December 2017, an outbreak of diphtheria occurred in Indonesia, with more than 600 cases found and 38 fatalities.[77]
In November 2019, two cases of diphtheria occurred in the Lothian area of Scotland.[78] Additionally, in November 2019, an unvaccinated 8-year-old boy died of diphtheria in Athens, Greece.[79]
In July 2022, two cases of diphtheria occurred in northern New South Wales, Australia.[80]
In October 2022, there was an outbreak of diphtheria at the former Manston airfield, a former Ministry of Defence (MoD) site in Kent, England, which had been converted to an asylum seeker processing centre. The capacity of the processing centre was 1,000 people, although about 3,000 were living at the site, with some accommodated in tents. The Home Office, the government department responsible for asylum seekers, refused to confirm the number of cases.[81]
In December 2023 there was an outbreak at a school in Luton, in the United Kingdom. UK Health Security Agency (UKHSA) issued a statement saying specialists have been providing public health support following confirmation of the diphtheria case at a primary school in Luton. The agency said it is working closely with local and national partners "to ensure all necessary public health measures are implemented" following the discovery of the new case. The statement added: "We have conducted a risk assessment and close contacts of the case have been identified and where appropriate, vaccination and advice will be given to prevent the spread of the infection."[82]