| |
 | |
| Names | |
|---|---|
| IUPAC name
Lithium tantalate
| |
| Other names
Lithium metatantalate
| |
| Identifiers | |
| ECHA InfoCard | 100.031.584 |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| LiTaO3 | |
| Molar mass | 235.887 g/mol |
| Density | 7.46 g/cm3, solid |
| Melting point | 1,650 °C (3,000 °F; 1,920 K) |
| Insoluble in water | |
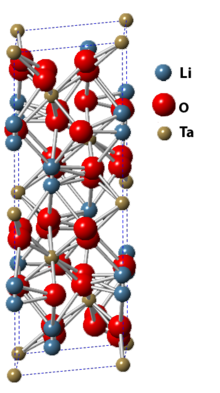
| Structure | |
| Space group R3c | |
a = 515.43 pm, c = 1378.35 pm[1]
| |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Acute Toxicity: Oral, Inhalation, Dermal |
| Safety data sheet (SDS) | http://www.samaterials.com/pdf/Lithium-Tantalate-Wafers-(LiTaO3-Wafers)-sds.pdf |
| Related compounds | |
Other anions
|
LiNbO3 |
| Supplementary data page | |
| Lithium tantalate (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium tantalate is the inorganic compound with the formula LiTaO3. It is a white, diamagnetic, water-insoluble solid. The compound has the perovskite structure. It has optical, piezoelectric, and pyroelectric properties. Considerable information is available from commercial sources about this material.[2]
Lithium tantalate is produced by treating tantalum(V) oxide with lithium oxide. The use of excess alkali gives water-soluble polyoxotantalates. Single crystals of Lithium tantalate are pulled from the melt using the Czochralski method.[2]
Lithium tantalate is used for nonlinear optics, passive infrared sensors such as motion detectors, terahertz generation and detection, surface acoustic wave applications, cell phones. Lithium tantalate is a standard detector element in infrared spectrophotometers.[3]
The phenomenon of pyroelectric fusion has been demonstrated using a lithium tantalate crystal producing a large enough charge to generate and accelerate a beam of deuterium nuclei into a deuterated target resulting in the production of a small flux of helium-3 and neutrons through nuclear fusion without extreme heat or pressure.[4]
A difference between positively and negatively charged parts of pyroelectric LiTaO3 crystals was observed when water freezes to them.[5]