Chemical compound
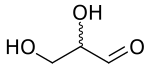
Glyceraldehyde (glyceral) is a triose monosaccharide with chemical formula C3H6O3. It is the simplest of all common aldoses. It is a sweet, colorless, crystalline solid that is an intermediate compound in carbohydrate metabolism. The word comes from combining glycerol and aldehyde, as glyceraldehyde is glycerol with one alcohol group oxidized to an aldehyde.
Structure
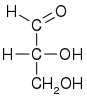
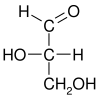
Glyceraldehyde has one chiral center and therefore exists as two different enantiomers with opposite optical rotation:
- In the D/L nomenclature, either D from Latin Dexter meaning "right", or L from Latin Laevo meaning "left"
- In the R/S nomenclature, either R from Latin Rectus meaning "right", or S from Latin Sinister meaning "left"
While the optical rotation of glyceraldehyde is (+) for R and (−) for S, this is not true for all monosaccharides. The stereochemical configuration can only be determined from the chemical structure, whereas the optical rotation can only be determined empirically (by experiment).
It was by a lucky guess that the molecular D- geometry was assigned to (+)-glyceraldehyde in the late 19th century, as confirmed by X-ray crystallography in 1951.[2]
Nomenclature
In the D/L system, glyceraldehyde is used as the configurational standard for carbohydrates.[3] Monosaccharides with an absolute configuration identical to (R)-glyceraldehyde at the last stereocentre, for example C5 in glucose, are assigned the stereo-descriptor D-. Those similar to (S)-glyceraldehyde are assigned an L-.
Chemical synthesis
Glyceraldehyde can be prepared, along with dihydroxyacetone, by the mild oxidation of glycerol, for example with hydrogen peroxide[4] and a ferrous salt as catalyst.[citation needed]
Its cyclohexylidene acetal can also be produced by oxidative cleavage of the bis(acetal) of mannitol.[5]
Biochemistry
The enzyme glycerol dehydrogenase (NADP+) has two substrates, glycerol and NADP+, and 3 products, D-glyceraldehyde, NADPH and H+.[6]
The interconversion of the phosphates of glyceraldehyde (glyceraldehyde 3-phosphate) and dihydroxyacetone (dihydroxyacetone phosphate), catalyzed by the enzyme triosephosphate isomerase, is an intermediate step in glycolysis.