 | |
| Clinical data | |
|---|---|
| Trade names | Livostin |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | Ophthalmic, intranasal[1] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
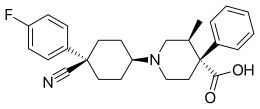
| Formula | C26H29FN2O2 |
| Molar mass | 420.528 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Levocabastine (trade name Livostin or Livocab, depending on the region) is a selective second-generation H1 receptor antagonist which was discovered at Janssen Pharmaceutica in 1979. It is used for allergic conjunctivitis.[3]
As well as acting as an antihistamine, levocabastine has also subsequently been found to act as a potent and selective antagonist for the neurotensin receptor NTS2, and was the first drug used to characterise the different neurotensin subtypes.[4][5] This has made it a useful tool for the study of this receptor.[6][7][8][9][10]
The pharmaceutical drug Bilina is a combination of Levocabastine, benzalkonium chloride, and other components and is typically used in a 0.5 mg/ml suspension as eye-drops, dispensed in 4ml bottles for the treatment of allergic conjunctivitis or similar allergic ocular conditions.[11]