| |
| Names | |
|---|---|
| IUPAC name
Niobium(V) oxide
| |
| Other names
Niobium pentoxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.013.831 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| Nb2O5 | |
| Molar mass | 265.81 g/mol |
| Appearance | white orthogonal solid |
| Density | 4.60 g/cm3 |
| Melting point | 1,512 °C (2,754 °F; 1,785 K) |
| insoluble | |
| Solubility | soluble in HF |
| -10·10−6 cm3/mol [1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Niobium pentoxide is the inorganic compound with the formula Nb2O5. A colorless, insoluble, and fairly unreactive solid, it is the most widespread precursor for other compounds and materials containing niobium. It is predominantly used in alloying, with other specialized applications in capacitors, optical glasses, and the production of lithium niobate.[2]
Structure
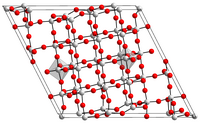
[edit]It has many polymorphic forms all based largely on octahedrally coordinated niobium atoms.[3][4] The polymorphs are identified with a variety of prefixes.[3][4] The form most commonly encountered is monoclinic H-Nb2O5, which has a complex structure with a unit cell containing 28 niobium atoms and 70 oxygen, where 27 of the niobium atoms are octahedrally coordinated and one tetrahedrally.[5] There is an uncharacterised solid hydrate, Nb2O5·nH2O, the so-called niobic acid (previously called columbic acid), which can be prepared by hydrolysis of a basic solution of niobium pentachloride or Nb2O5 dissolved in HF.[6]
Molten niobium pentoxide has lower mean coordination numbers than the crystalline forms, with a structure comprising mostly NbO5 and NbO6 polyhedra.[7]
Production
[edit]Hydrolysis
[edit]Nb2O5 is prepared by hydrolysis of alkali-metal niobates, alkoxides or fluoride using base. Such ostensibly simple procedures afford hydrated oxides that can then be calcined. Pure Nb2O5 can also be prepared by hydrolysis of NbCl5:[8]
- 2 NbCl5 + 5 H2O → Nb2O5 + 10 HCl
A method of production via sol-gel techniques has been reported hydrolysing niobium alkoxides in the presence of acetic acid, followed by calcination of the gels to produce the orthorhombic form,[3] T-Nb2O5.[9]
Oxidation
[edit]Given that Nb2O5 is the most common and robust compound of niobium, many methods, both practical and esoteric, exist for its formation. The oxide for example, arises when niobium metal is oxidised in air.[10] The oxidation of niobium dioxide, NbO2 in air forms the polymorph, L-Nb2O5.[11]
Nano-sized niobium pentoxide particles have been synthesized by LiH reduction of NbCl5, followed by aerial oxidation as part of a synthesis of nano structured niobates.[citation needed]
Reactions
[edit]Nb2O5 is attacked by HF and dissolves in fused alkali.[6][10]
Reduction to the metal
[edit]The conversion of Nb2O5 is the main route for the industrial production of niobium metal. In the 1980s, about 15,000,000 kg of Nb2O5 were consumed annually for reduction to the metal.[12] The main method is reduction of this oxide with aluminium:
- 3 Nb2O5 + 10 Al → 6 Nb + 5 Al2O3
An alternative but less practiced route involves carbothermal reduction, which proceeds via reduction with carbon and forms the basis of the two stage Balke process:[13][14]
- Nb2O5 + 7 C → 2 NbC + 5 CO (heated under vacuum at 1800 °C)
- 5 NbC + Nb2O5 → 7 Nb + 5 CO
Conversion to halides
[edit]Many methods are known for conversion of Nb2O5 to the halides. The main problem is incomplete reaction to give the oxyhalides. In the laboratory, the conversion can be effected with thionyl chloride:[15]
- Nb2O5 + 5 SOCl2 → 2 NbCl5 + 5 SO2
Nb2O5 reacts with CCl4 to give niobium oxychloride NbOCl3.
Conversion to niobates
[edit]Treating Nb2O5 with aqueous NaOH at 200 °C can give crystalline sodium niobate, NaNbO3 whereas the reaction with KOH may yield soluble Lindqvist-type hexaniobates, Nb
6O8−
19.[16] Lithium niobates such as LiNbO3 and Li3NbO4 can be prepared by reaction lithium carbonate and Nb2O5.[17][18]
Conversion to reduced niobium oxides
[edit]High temperature reduction with H2 gives NbO2:[10]
- Nb2O5 + H2 → 2 NbO2 + H2O
Niobium monoxide arises from a comproportionation using an arc-furnace:[19]
- Nb2O5 + 3Nb → 5 NbO
The burgundy-coloured niobium(III) oxide, one of the first superconducting oxides, can be prepared again by an comproportionation:[18]
- Li3NbO4 + 2 NbO → 3 LiNbO2
Uses
[edit]Niobium pentoxide is used mainly in the production of niobium metal,[12] but specialized applications exist in the production of optical glasses and lithium niobate.[2]
Thin films of Nb2O5 form the dielectric layers in niobium electrolytic capacitors.
Nb2O5 have been considered for use as an anode in a lithium-ion battery, given that their ordered crystalline structure allows charging speeds of 225 mAh g−1 at 200 mA g−1 across 400 cycles, at a Coulombic efficiency of 99.93%.[20]
External links
[edit]References
[edit]- ^ "Handbook of Chemistry and Physics 102nd Edition". CRC Press.
- ^ a b Francois Cardarelli (2008) Materials Handbook Springer London ISBN 978-1-84628-668-1
- ^ a b c C. Nico; et al. (2011). "Sintered NbO powders for electronic device applications". The Journal of Physical Chemistry C. 115 (11): 4879–4886. doi:10.1021/jp110672u.
- ^ a b Wells A.F. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6
- ^ Gatehouse, B. M.; Wadsley, A. D. (1964-12-01). "The crystal structure of the high temperature form of niobium pentoxide". Acta Crystallographica. 17 (12). International Union of Crystallography (IUCr): 1545–1554. doi:10.1107/s0365110x6400384x. ISSN 0365-110X.
- ^ a b D.A. Bayot and M.M. Devillers, Precursors routes for the preparation of Nb based multimetallic oxides in Progress in Solid State Chemistry Research, Arte M. Newman, Ronald W. Buckley, (2007),Nova Publishers, ISBN 1-60021-313-8
- ^ Alderman, O. L. G. Benmore, C. J. Neuefeind, J. C. Coillet, E Mermet, Alain Martinez, V. Tamalonis, A. Weber, J. K. R. (2018). "Amorphous tantala and its relationship with the molten state". Physical Review Materials. 2 (4): 043602. Bibcode:2018PhRvM...2d3602A. doi:10.1103/PhysRevMaterials.2.043602.
((cite journal)): CS1 maint: multiple names: authors list (link) - ^ Process for the manufacture of niobium pentoxide or tantalum pentoxide, Kern, Therwil, Jacob, Hooper (CIBA Switzerland), US Patent number: 3133788, (1964)
- ^ Griesmar, P.; Papin, G.; Sanchez, C.; Livage, J. (1991). "Sol-gel route to niobium pentoxide". Chemistry of Materials. 3 (2). American Chemical Society (ACS): 335–339. doi:10.1021/cm00014a026. ISSN 0897-4756.
- ^ a b c Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Vezzoli, G. C. (1982-10-01). "Electrical properties of NbO2andNb2O5at elevated temperature in air and flowing argon". Physical Review B. 26 (7). American Physical Society (APS): 3954–3957. Bibcode:1982PhRvB..26.3954V. doi:10.1103/physrevb.26.3954. ISSN 0163-1829.
- ^ a b Albrecht, Sven; Cymorek, Christian; Eckert, Joachim (2011), Ullmann's Encyclopedia of Industrial Chemistry: Niobium and Niobium Compounds, Weinheim: Wiley-VCH, doi:10.1002/14356007.a17_251.pub2
- ^ Alan E. Comyns (1999) Encyclopedic Dictionary of Named Processes in Chemical Technology CRC Press, ISBN 0-8493-1205-1
- ^ U.S. Environmental Protection Agency, Development Document for Effluent Limitations, Guidelines and Standards for the Nonferrous Metals Manufacturing Point Source Category, Volume VIII, Office of Water Regulations and Standards, May 1989
- ^ Brown, D. (1967). "Niobium(V) Chloride and Hexachloroniobates(V)". Inorganic Syntheses. Inorganic Syntheses. Vol. 9. pp. 88–92. doi:10.1002/9780470132401.ch24. ISBN 9780470132401.
- ^ Santos, I.C.M.S.; Loureiro, L.H.; Silva, M.F.P.; Cavaleiro, Ana M.V. (2002). "Studies on the hydrothermal synthesis of niobium oxides". Polyhedron. 21 (20). Elsevier BV: 2009–2015. doi:10.1016/s0277-5387(02)01136-1. ISSN 0277-5387.
- ^ US Patent 5482001 - Process for producing lithium niobate single crystal,1996, Katoono T., Tominaga H.,
- ^ a b Geselbracht, Margret J.; Stacy, Angelica M.; Rosseinsky, Matthew (2007-01-05). "Lithium Niobium Oxide: LiNbO2 and Superconducting LixNbO2". Inorganic Syntheses. Vol. 30. Hoboken, NJ, USA: John Wiley & Sons, Inc. pp. 222–226. doi:10.1002/9780470132616.ch42. ISBN 9780470132616. ISSN 1934-4716.
- ^ Reed, T. B.; Pollard, E. R.; Lonney, L. E.; Loehman, R. E.; Honig, J. M. (2007-01-05). "Niobium Monoxide". Inorganic Syntheses. Vol. 30. Hoboken, NJ, USA: John Wiley & Sons, Inc. pp. 108–110. doi:10.1002/9780470132616.ch22. ISBN 9780470132616. ISSN 1934-4716.
- ^ Lavars, Nick (2022-09-09). "Battery electrode transforms during use for faster charging". New Atlas. Retrieved 2022-09-10.
| Niobium(II) | |||
|---|---|---|---|
| Niobium(III) | |||
| Niobium(IV) | |||
| Niobium(V) |
| ||