| |
| |
| Names | |
|---|---|
| IUPAC name
cobalt(II) dicobalt(III) oxide
| |
| Other names
cobalt oxide, cobalt(II,III) oxide, cobaltosic oxide, tricobalt tetroxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.013.780 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Co3O4 CoO.Co2O3 | |
| Molar mass | 240.80 g/mol |
| Appearance | black solid |
| Density | 6.07 g/cm3[2] |
| Melting point | 895 °C (1,643 °F; 1,168 K) |
| Boiling point | 900 °C (1,650 °F; 1,170 K) (decomposes) |
| Insoluble | |
| Solubility | soluble (with degradation) in acids and alkalis |
| +7380·10−6 cm3/mol | |
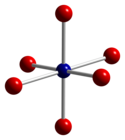
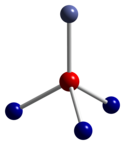
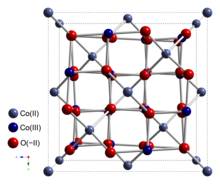
| Structure | |
| cubic | |
| Fd3m, No. 227[3] | |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H317, H334, H350, H411 | |
| P261, P273, P284, P304+P340, P342+P311 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cobalt(II,III) oxide is an inorganic compound with the formula Co3O4. It is one of two well characterized cobalt oxides. It is a black antiferromagnetic solid. As a mixed valence compound, its formula is sometimes written as CoIICoIII2O4 and sometimes as CoO•Co2O3.[4]