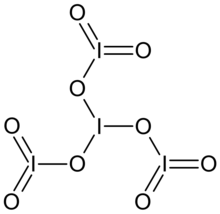
Tetraiodine nonoxide
|
| Identifiers
|
|
|
|
|
|
|
InChI=1S/I4O9/c5-1(6)11-4(12-2(7)8)13-3(9)10 Key: IQQBBNVVZPXURM-UHFFFAOYSA-N
|
O=I(=O)OI(OI(=O)=O)OI(=O)=O
|
| Properties
|
|
|
I4O9
|
| Molar mass
|
651.609 g·mol−1
|
| Appearance
|
light yellow solid[1]
|
| Melting point
|
75 °C (decomposes)[1]
|
|
|
reacts
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
Chemical compound
Tetraiodine nonoxide is an iodine oxide with the chemical formula I4O9.
Preparation
Tetraiodine nonoxide can be produced by reacting ozone and iodine in carbon tetrachloride at −78 °C:[2][3]
- 2 I2 + 9 O3 → I4O9 + 9 O2
It can also be produced by heating iodic acid and phosphoric acid together:[4]
- 8 HIO3 → 2 I4O9 + 4 H2O + O2
Properties
Tetraiodine nonoxide is a light yellow solid that can easily hydrolyze. It decomposes above 75 °C:[2]
- 4 I4O9 → 6 I2O5 + 2 I2 + 3 O2
Like diiodine tetroxide, tetraiodine nonoxide contains both I(III) and I(V), and disproportionate to iodate and iodide under alkaline conditions:[2]
- 3 I4O9 + 12 HO− → I− + 11 IO−3 + 6 H2O
It reacts with water to form iodic acid and iodine:[3]
- 4 I4O9 + 9 H2O → 18 HIO3 + I2
