| |
| Names | |
|---|---|
| Other names
Yuremamine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
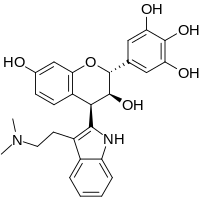
| C27H28N2O6 | |
| Molar mass | 476.529 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Yuremamine is a phytoindole alkaloid which was isolated from the bark of Mimosa tenuiflora in 2005, and erroneously assigned a pyrrolo[1,2-a]indole structure that was thought to represent a new class of indole alkaloids.[2] However, in 2015, the bioinspired total synthesis of yuremamine revealed its structure to be a flavonoid derivative.[3] It was also noted in the original isolation of yuremamine that the alkaloid occurs naturally as a purple solid, but total synthesis revealed that yuremamine as a free base is colorless, and the formation of a trifluoroacetate salt during HPLC purification is what led to the purple appearance.[3]

Originally proposed chemical structure of yuremamine