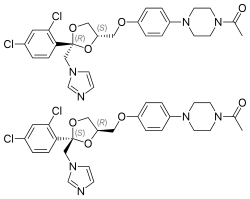
 (2R,4S)-(+)-ketoconazole (top) (2S,4R)-(−)-ketoconazole (bottom) | |
 Ball-and-stick model of (2R,4S)-(+)-ketoconazole | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌkiːtoʊˈkoʊnəˌzoʊl, -zɒl/[1][2] |
| Trade names | Nizoral, others |
| Other names | R-41400; KW-1414 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682816 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (tablets), topical (cream, shampoo, solution) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | By mouth: 37–97%[8] |
| Protein binding | 84 to 99% |
| Metabolism | Extensive liver (predominantly oxidation, O-dealkylation) |
| Metabolites | N-deacetyl ketoconazole |
| Elimination half-life | Biphasic |
| Excretion | Bile duct (major) and kidney[9] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.059.680 |
| Chemical and physical data | |
| Formula | C26H28Cl2N4O4 |
| Molar mass | 531.43 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture[9][10] |
| |
| |
| | |

Ketoconazole, sold under the brand name Nizoral among others, is an antiandrogen, antifungal, and antiglucocorticoid medication used to treat a number of fungal infections.[11] Applied to the skin it is used for fungal skin infections such as tinea, cutaneous candidiasis, pityriasis versicolor, dandruff, and seborrheic dermatitis.[12] Taken by mouth it is a less preferred option and only recommended for severe infections when other agents cannot be used.[11] Other uses include treatment of excessive male-patterned hair growth in women and Cushing's syndrome.[11]
Common side effects when applied to the skin include redness.[12] Common side effects when taken by mouth include nausea, headache, and liver problems.[11] Liver problems may result in death or the need for a liver transplantation.[11][13] Other severe side effects when taken by mouth include QT prolongation, adrenocortical insufficiency, and anaphylaxis.[11][13] It is an imidazole and works by hindering the production of ergosterol required for the fungal cell membrane, thereby slowing growth.[11]
Ketoconazole was patented in 1977 by Belgian pharmaceutical company Janssen, and came into medical use in 1981.[14] It is available as a generic medication and formulations that are applied to the skin are over the counter in the United Kingdom.[12] In 2021, it was the 161st most commonly prescribed medication in the United States, with more than 3 million prescriptions.[15][16] The formulation that is taken by mouth was withdrawn in the European Union and in Australia in 2013,[17][18] and in China in 2015.[19] In addition, its use was restricted in the United States and Canada in 2013.[18]
Medical uses
[edit]Topical antifungal
[edit]Topically administered ketoconazole is usually prescribed for fungal infections of the skin and mucous membranes, such as athlete's foot, ringworm, candidiasis (yeast infection or thrush), jock itch, and tinea versicolor.[20] Topical ketoconazole is also used as a treatment for dandruff (seborrheic dermatitis of the scalp) and for seborrheic dermatitis on other areas of the body, perhaps acting in these conditions by suppressing levels of the fungus Malassezia furfur on the skin.[20][21][22]
Systemic antifungal
[edit]Ketoconazole has activity against many kinds of fungi that may cause human disease, such as Candida, Histoplasma, Coccidioides, and Blastomyces (although it is not active against Aspergillus), chromomycosis and paracoccidioidomycosis.[23][13] First made in 1977,[20] ketoconazole was the first orally-active azole antifungal medication.[23] However, ketoconazole has largely been replaced as a first-line systemic antifungal medication by other azole antifungal agents, such as fluconazole and/or itraconazole, because of ketoconazole's greater toxicity, poorer absorption, and more limited spectrum of activity.[23][24]
Ketoconazole is used orally in dosages of 200 to 400 mg per day in the treatment of superficial and deep fungal infections.[25]
Off-label uses
[edit]Hair loss
[edit]
Ketoconazole shampoo in conjunction with an oral 5α-reductase inhibitor such as finasteride or dutasteride has been used off label to treat androgenic alopecia. It was speculated that antifungal properties of ketoconazole reduce scalp microflora and consequently may reduce follicular inflammation that contributes to alopecia.[26]
Limited clinical studies suggest ketoconazole shampoo used either alone[27][28] or in combination with other treatments[29] may be useful in reducing hair loss in some cases.[30]
Hormonal
[edit]The side effects of ketoconazole are sometimes harnessed in the treatment of non-fungal conditions. While ketoconazole blocks the synthesis of the sterol ergosterol in fungi, in humans, at high dosages (>800 mg/day), it potently inhibits the activity of several enzymes necessary for the conversion of cholesterol to steroid hormones such as testosterone and cortisol.[23][25] Specifically, ketoconazole has been shown to inhibit cholesterol side-chain cleavage enzyme, which converts cholesterol to pregnenolone, 17α-hydroxylase and 17,20-lyase,[25] which convert pregnenolone into androgens, and 11β-hydroxylase, which converts 11-deoxycortisol to cortisol.[31] All of these enzymes are mitochondrial cytochrome p450 enzymes.[32] Based on these antiandrogen and antiglucocorticoid effects, ketoconazole has been used with some success as a second-line treatment for certain forms of advanced prostate cancer[25][33] and for the suppression of glucocorticoid synthesis in the treatment of Cushing's syndrome.[34] However, in the treatment of prostate cancer, concomitant glucocorticoid administration is needed to prevent adrenal insufficiency.[25] Ketoconazole has additionally been used, in lower dosages, to treat hirsutism and, in combination with a GnRH analogue, male-limited precocious puberty.[25] In any case, the risk of hepatotoxicity with ketoconazole limits its use in all of these indications, especially in those that are benign such as hirsutism.[25]
Ketoconazole has been used to prevent the testosterone flare at the initiation of GnRH agonist therapy in men with prostate cancer.[35]
Contraindications
[edit]Oral ketoconazole has various contraindications, such as concomitant use with certain other drugs due to known drug interactions.[6] Other contraindications of oral ketoconazole include liver disease, adrenal insufficiency, and known hypersensitivity to oral ketoconazole.[6]
Side effects
[edit]Gastrointestinal
[edit]Vomiting, diarrhea, nausea, constipation, abdominal pain, upper abdominal pain, dry mouth, dysgeusia, dyspepsia, flatulence, tongue discoloration may occur.[36]
Endocrine
[edit]The drug may cause adrenal insufficiency so the level of the adrenocortical hormones should be monitored while taking it.[13][36] Oral ketoconazole at a dosage range of 400 to 2,000 mg/day has been found to result in a rate of gynecomastia of 21%.[37]
Liver
[edit]In July 2013, the US Food and Drug Administration (FDA) issued a warning that taking ketoconazole by mouth can cause severe liver injuries and adrenal gland problems: adrenal insufficiency and worsening of other related to the gland conditions.[13] It recommends oral tablets should not be a first-line treatment for any fungal infection. It should be used for the treatment of certain fungal infections, known as endemic mycoses, only when alternative antifungal therapies are not available or tolerated.[13] As contraindication it should not be used in people with acute or chronic liver disease.[13]
Hypersensitivity
[edit]Anaphylaxis after the first dose may occur.[medical citation needed] Other cases of hypersensitivity include urticaria.[11][6]
Topical formulations
[edit]The topical formulations have not been associated with liver damage, adrenal problems, or drug interactions. These formulations include creams, shampoos, foams, and gels applied to the skin, unlike the ketoconazole tablets, which are taken by mouth.[13]
Pregnancy
[edit]Ketoconazole is categorized as pregnancy category C in the US.[38] Research in animals has shown it to cause teratogenesis when administered in high doses.[38] A subsequent trial in Europe failed to show a risk to infants of mothers receiving ketoconazole.[39]
Overdose
[edit]In the event of an overdose of oral ketoconazole, treatment should be supportive and based on symptoms.[6] Activated charcoal may be administered within the first hour following overdose of oral ketoconazole.[6]
Interactions
[edit]The concomitant use of the following medications is contraindicated with ketoconazole tablets:[6][36]
- methadone, disopyramide, dronedarone
- irinotecan, lurasidone, colchicine
- alprazolam, oral midazolam, oral triazolam
- felodipine, ranolazine, tolvaptan, eplerenone
- HMG-CoA reductase inhibitors: lovastatin, simvastatin
- ergot alkaloids: ergotamine, dihydroergotamine, ergometrine, methylergometrine
- Others: cisapride, nisoldipine, dofetilide, pimozide
And is not recommended:[6][36]
- carbamazepine, phenytoin
- gastric acid suppressants: antacids, antimuscarinics, histamine H2 blockers, proton pump inhibitors
- sucralfate
- rifampin, rifabutin, isoniazid
- efavirenz, nevirapine
Ritonavir is known for increasing activity of the ketoconazole so it is recommended to reduce dosage.[6]
There is also a list of drugs which significantly decrease systemic exposure to the ketoconazole and drugs whose systemic exposure is increased by the ketoconazole.[6][36]
Pharmacology
[edit]Pharmacodynamics
[edit]Antifungal activity
[edit]As an antifungal, ketoconazole is structurally similar to imidazole, and interferes with the fungal synthesis of ergosterol, a constituent of fungal cell membranes, as well as certain enzymes. As with all azole antifungal agents, ketoconazole works principally by inhibiting the enzyme cytochrome P450 14α-demethylase (CYP51A1).[32] This enzyme participates in the sterol biosynthesis pathway that leads from lanosterol to ergosterol. Lower doses of fluconazole and itraconazole are required to kill fungi compared to ketoconazole, as they have been found to have a greater affinity for fungal cell membranes.
Resistance to ketoconazole has been observed in a number of clinical fungal isolates, including Candida albicans. Experimentally, resistance usually arises as a result of mutations in the sterol biosynthesis pathway. Defects in the sterol 5-6 desaturase enzyme reduce the toxic effects of azole inhibition of the 14-alpha demethylation step. Multidrug-resistance (MDR) genes can also play a role in reducing cellular levels of the drug. As azole antifungals all act at the same point in the sterol pathway, resistant isolates are normally cross-resistant to all members of the azole family.[40][41]
Antihormonal activity
[edit]As an antiandrogen, ketoconazole operates through at least two mechanisms of action. First, and most notably, high oral doses of ketoconazole (e.g. 40 mg three times per day) block both testicular and adrenal androgen biosynthesis, leading to a reduction in circulating testosterone levels.[25][42] It produces this effect through inhibition of 17α-hydroxylase and 17,20-lyase, which are involved in the synthesis and degradation of steroids, including the precursors of testosterone.[25] Due to its efficacy at reducing systemic androgen levels, ketoconazole has been used with some success as a treatment for androgen-dependent prostate cancer.[43] Second, ketoconazole is an androgen receptor antagonist, competing with androgens such as testosterone and dihydrotestosterone (DHT) for binding to the androgen receptor. This effect is thought to be quite weak however, even with high oral doses of ketoconazole.[44]
Ketoconazole, along with miconazole, has been found to act as an antagonist of the glucocorticoid receptor.[45][46]
Ketoconazole is a racemic mixture consisting of cis-(2S,4R)-(−) and cis-(2R,4S)-(+) enantiomers.[10] The cis-(2S,4R) isomer was more potent in inhibiting progesterone 17α,20-lyase than its enantiomer (IC50 values of 0.05 and 2.38 μM, respectively) and in inhibiting 11β-hydroxylase (IC50 values of 0.152 and 0.608 μM, respectively). Both isomers were relatively weak inhibitors of human placental aromatase.[9]
Oral ketoconazole has been used clinically as a steroidogenesis inhibitor in men, women, and children at dosages of 200 to 1,200 mg/day.[47][48][49] Numerous small studies have investigated the effects of oral ketoconazole on hormone levels in humans.[50] It has been found in men to significantly decrease testosterone and estradiol levels and to significantly increase luteinizing hormone, progesterone, and 17α-hydroxyprogesterone levels, whereas levels of androstenedione, follicle-stimulating hormone, and prolactin were unaffected.[50][51][48] The ratio of testosterone to estradiol is also decreased during oral ketoconazole therapy in men.[48] Suppression of testosterone levels by ketoconazole is generally partial and has often been found to be transient.[50] Better effects on suppression of testosterone levels have been observed in men when ketoconazole is combined with a GnRH agonist to suppress the hypothalamic–pituitary–gonadal axis, which prevents compensatory upregulation of luteinizing hormone secretion and consequent activation of gonadal testosterone production.[48] In premenopausal women with polycystic ovary syndrome, ketoconazole has been found to significantly decrease levels of androstenedione and testosterone and significantly increase levels of 17α-hydroxyprogesterone and estradiol.[49][52] Studies in postmenopausal women with breast cancer have found that ketoconazole significantly decreases androstenedione levels, slightly decreases estradiol levels, and does not affect estrone levels.[53] This indicates minimal inhibition of aromatase by ketoconazole in vivo in humans.[53] Ketoconazole has also been found to decrease levels of endogenous corticosteroids, such as cortisol, corticosterone, and aldosterone, as well as vitamin D.[54][48]
Ketoconazole has been found to displace dihydrotestosterone and estradiol from sex hormone-binding globulin in vitro, but this was not found to be relevant in vivo.[48]
Other activities
[edit]Ketoconazole has been found to inhibit the activity of the cation channel TRPM5.[55]
Pharmacokinetics
[edit]When administered orally, ketoconazole is best absorbed at highly acidic levels, so antacids or other causes of decreased stomach acid levels will lower the drug's absorption. Absorption can be increased by taking it with an acidic beverage, such as cola.[56] Ketoconazole is very lipophilic and tends to accumulate in fatty tissues.
Chemistry
[edit]Ketoconazole is a synthetic imidazole.[57][58] It is a nonsteroidal compound.[57][58] It is a racemic mixture of two enantiomers, levoketoconazole ((2S,4R)-(−)-ketoconazole) and dextroketoconazole ((2R,4S)-(+)-ketoconazole).[57][58] Levoketoconazole is under development for potential clinical use as a steroidogenesis inhibitor with better tolerability and less toxicity than ketoconazole. Other steroidogenesis inhibitors besides ketoconazole and levoketoconazole include the nonsteroidal compound aminoglutethimide and the steroidal compound abiraterone acetate.[citation needed]
History
[edit]Ketoconazole was discovered in 1976 at Janssen Pharmaceuticals.[59] It was patented in 1977,[14] followed by introduction in the United States in July 1981.[18][8][60][14] Following its introduction, ketoconazole was the only systemic antifungal available for almost a decade.[18] Ketoconazole was introduced as the prototypical medication of the imidazole group of antifungals.[61] Oral ketoconazole has been replaced with oral fluconazole or itraconazole for many mycoses.[61]
Due to incidence of serious liver toxicity, the use of oral ketoconazole was suspended in France in July 2011, following review.[18] This event triggered an evaluation of oral ketoconazole throughout the rest of the European Union.[18][62] In 2013, oral ketoconazole was withdrawn in the European Union and Australia, and strict restrictions were placed on the use of oral ketoconazole in the United States and Canada.[18] Oral ketoconazole is indicated for use in these countries when the indication is a severe or life-threatening systemic infection and alternatives are unavailable.[18] However, topical ketoconazole, which does not distribute systemically, is safe and widely used still.[18]
Ketoconazole HRA was approved for use in the European Union for treatment of Cushing's syndrome in November 2013.[7][63]
Society and culture
[edit]Generic names
[edit]Ketoconazole is the generic name of the drug and its INN, USAN, BAN, and JAN.[57][58][64][65]
Brand names
[edit]Ketoconazole has been marketed under a large number of brand names.[57][58][64][65]
Availability
[edit]Ketoconazole is available widely throughout the world.[58][65]
In 2013, the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) recommended that a ban be imposed on the use of oral ketoconazole for systemic use in humans throughout the European Union, after concluding that the risk of serious liver injury from systemic ketoconazole outweighs its benefits.[66]
Research
[edit]As of March 2019, oral levoketoconazole (developmental code name COR-003, tentative brand name Recorlev) is phase III clinical trials for the treatment of Cushing's syndrome.[67] Oral levoketoconazole may have a lower risk of liver toxicity than oral ketoconazole.[68]
Veterinary use
[edit]Ketoconazole is sometimes prescribed as an antifungal by veterinarians for use in pets, often as unflavored tablets that may need to be cut to smaller size for correct dosage.[69]
References
[edit]- ^ "Ketoconazole". Merriam-Webster.com Dictionary.
- ^ "Ketoconazole". Dictionary.com Unabridged (Online). n.d.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Ketoconazole HRA 200mg Tablets - Summary of Product Characteristics (SmPC)". (emc). 18 September 2017. Archived from the original on 2 August 2020. Retrieved 1 April 2020.
- ^ "Ketoconazole 2% w/w Shampoo - Summary of Product Characteristics (SmPC)". (emc). 5 October 2015. Archived from the original on 3 August 2020. Retrieved 1 April 2020.
- ^ a b c d e f g h i j "Ketoconazole tablet". DailyMed. 26 June 2018. Archived from the original on 5 August 2020. Retrieved 5 January 2020.
- ^ a b "Ketoconazole HRA EPAR". European Medicines Agency (EMA). 23 April 2012. Archived from the original on 3 August 2020. Retrieved 1 April 2020.
- ^ a b Millikan LE (19 April 2016). Drug Therapy in Dermatology. CRC Press. pp. 82–. ISBN 978-0-203-90831-0.
- ^ a b c "Assessment report: Ketoconazole HRA" (PDF). European Medicines Agency (EMA). Archived (PDF) from the original on 27 August 2016. Retrieved 26 August 2016.
- ^ a b Arakaki R, Welles B (February 2010). "Ketoconazole enantiomer for the treatment of diabetes mellitus". Expert Opinion on Investigational Drugs. 19 (2): 185–94. doi:10.1517/13543780903381411. PMID 20047506. S2CID 26531459.
- ^ a b c d e f g h "Ketoconazole Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 28 December 2010. Retrieved 23 March 2019.
- ^ a b c British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1198. ISBN 9780857113382.
- ^ a b c d e f g h "FDA limits usage of Nizoral (ketoconazole) oral tablets due to potentially fatal liver injury and risk of drug interactions and adrenal gland problems". FDA Drug Safety Communication. U.S. Food and Drug Administration. 26 July 2013. Archived from the original on 2 December 2013. Retrieved 23 November 2013.
- ^ a b c Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 503. ISBN 9783527607495.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Ketoconazole - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 14 January 2024.
- ^ "Oral ketoconazole (Nizoral) 200 mg tablets". Therapeutic Goods Administration (TGA). 10 October 2013. Archived from the original on 2 July 2015. Retrieved 23 March 2019.
- ^ a b c d e f g h i Gupta AK, Lyons DC (2015). "The Rise and Fall of Oral Ketoconazole". Journal of Cutaneous Medicine and Surgery. 19 (4): 352–7. doi:10.1177/1203475415574970. PMID 25775613. S2CID 206695486.
- ^ "国家食品药品监督管理总局关于停止生产销售使用酮康唑口服制剂的公告(2015年第85号)" (in Chinese). China Food and Drug Administration. 25 June 2015. Archived from the original on 2 July 2015. Retrieved 2 July 2015.
- ^ a b c Phillips RM, Rosen T (2013). "Topical Antifungal Agents". In Wolverton SE (ed.). Comprehensive Dermatologic Therapy (3rd ed.). Philadelphia: Saunders. pp. 460–472. ISBN 978-1-4377-2003-7.
- ^ Neider R, Fritsch PO (2012). "Other Eczematous Eruptions". In Bolognia JL (ed.). Dermatology (3rd ed.). Philadelphia: Saunders. pp. 219–221. ISBN 9780723435716.
- ^ Young BK, Brodell RT, Cooper KD (2013). "Therapeutic Shampoos". In Wolverton SE (ed.). Comprehensive Dermatologic Therapy (3rd ed.). Philadelphia: Saunders. pp. 562–569. ISBN 978-1-4377-2003-7.
- ^ a b c d Finkel R, Cubeddu LX, Clark MA (2009). Pharmacology (4th ed.). Baltimore: Lippincott Williams & Wilkins. p. 411.
- ^ Kauffman CA (2004). "Introduction to the Mycoses". In Goldman L, Ausiello D (eds.). Cecil Textbook of Medicine (22nd ed.). Philadelphia: Saunders. p. 2043. ISBN 978-0-7216-9652-2.
- ^ a b c d e f g h i Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1197–. ISBN 978-0-7817-1750-2.
- ^ McElwee KJ, Shapiro JS (June 2012). "Promising therapies for treating and/or preventing androgenic alopecia". Skin Therapy Letter. 17 (6): 1–4. PMID 22735503. Archived from the original on 12 December 2015.
- ^ Piérard-Franchimont C, De Doncker P, Cauwenbergh G, Piérard GE (1998). "Ketoconazole shampoo: effect of long-term use in androgenic alopecia". Dermatology. 196 (4): 474–7. doi:10.1159/000017954. PMID 9669136. S2CID 30635892.
- ^ Piérard-Franchimont C, Goffin V, Henry F, Uhoda I, Braham C, Piérard GE (October 2002). "Nudging hair shedding by antidandruff shampoos. A comparison of 1% ketoconazole, 1% piroctone olamine and 1% zinc pyrithione formulations". International Journal of Cosmetic Science. 24 (5): 249–56. doi:10.1046/j.1467-2494.2002.00145.x. hdl:2268/11902. PMID 18498517. Archived from the original on 29 August 2021. Retrieved 4 July 2019.
- ^ Khandpur S, Suman M, Reddy BS (August 2002). "Comparative efficacy of various treatment regimens for androgenetic alopecia in men". The Journal of Dermatology. 29 (8): 489–98. doi:10.1111/j.1346-8138.2002.tb00314.x. PMID 12227482. S2CID 20886812.
- ^ Marks DH, Prasad S, De Souza B, Burns LJ, Senna MM (December 2019). "Topical Antiandrogen Therapies for Androgenetic Alopecia and Acne Vulgaris". Am J Clin Dermatol. 21 (2): 245–254. doi:10.1007/s40257-019-00493-z. PMID 31832993. S2CID 209331373.
- ^ "MedScape". Ectopic Cortisol Production Derived From Malignant Testicular Masses: Treatment and Management. Nature Publishing Group. Archived from the original on 13 May 2018. Retrieved 18 April 2015.
- ^ a b Loose DS, Kan PB, Hirst MA, Marcus RA, Feldman D (May 1983). "Ketoconazole blocks adrenal steroidogenesis by inhibiting cytochrome P450-dependent enzymes". The Journal of Clinical Investigation. 71 (5): 1495–9. doi:10.1172/JCI110903. PMC 437014. PMID 6304148.
- ^ Zelefsky MJ, Eastham JA, Sartor OA, Kantoff P (2008). DeVita VT, Lawrence TS, Rosenberg SA (eds.). Cancer: Principles & Practice of Oncology (8th ed.). Philadelphia: Lippincott Williams & Wilkins. p. 1443. ISBN 9780781772075.
- ^ Loli P, Berselli ME, Tagliaferri M (December 1986). "Use of ketoconazole in the treatment of Cushing's syndrome". The Journal of Clinical Endocrinology and Metabolism. 63 (6): 1365–71. doi:10.1210/jcem-63-6-1365. PMID 3023421.
- ^ Thompson IM (2001). "Flare Associated with LHRH-Agonist Therapy". Reviews in Urology. 3 (Suppl 3): S10–4. PMC 1476081. PMID 16986003.
- ^ a b c d e "Nizoral (Ketoconazole): Side Effects, Interactions, Warning, Dosage & Uses". RxList. Archived from the original on 7 April 2019. Retrieved 7 April 2019.
- ^ Deepinder F, Braunstein GD (September 2012). "Drug-induced gynecomastia: an evidence-based review". Expert Opinion on Drug Safety. 11 (5): 779–95. doi:10.1517/14740338.2012.712109. PMID 22862307. S2CID 22938364.
- ^ a b "Ketoconazole (Nizoral) Use During Pregnancy". Drugs.com. Archived from the original on 10 April 2020. Retrieved 24 May 2020.
- ^ Kazy Z, Puhó E, Czeizel AE (March 2005). "Population-based case-control study of oral ketoconazole treatment for birth outcomes". Congenital Anomalies. 45 (1): 5–8. doi:10.1111/j.1741-4520.2005.00053.x. PMID 15737124. S2CID 41187361.
- ^ Cartledge JD, Midgley J, Gazzard BG (December 1997). "Clinically significant azole cross-resistance in Candida isolates from HIV-positive patients with oral candidosis". AIDS. 11 (15): 1839–44. doi:10.1097/00002030-199715000-00008. PMID 9412702. S2CID 8440973.
- ^ Sanglard D, Ischer F, Monod M, Bille J (February 1997). "Cloning of Candida albicans genes conferring resistance to azole antifungal agents: characterization of CDR2, a new multidrug ABC transporter gene". Microbiology. 143 (Pt 2): 405–16. doi:10.1099/00221287-143-2-405. PMID 9043118.
- ^ Witjes FJ, Debruyne FM, Fernandez del Moral P, Geboers AD (May 1989). "Ketoconazole high dose in management of hormonally pretreated patients with progressive metastatic prostate cancer. Dutch South-Eastern Urological Cooperative Group". Urology. 33 (5): 411–5. doi:10.1016/0090-4295(89)90037-X. PMID 2652864.
- ^ De Coster R, Wouters W, Bruynseels J (January 1996). "P450-dependent enzymes as targets for prostate cancer therapy". The Journal of Steroid Biochemistry and Molecular Biology. 56 (1–6 Spec No): 133–43. doi:10.1016/0960-0760(95)00230-8. PMID 8603034. S2CID 42845713.
- ^ Eil C (August 1992). "Ketoconazole binds to the human androgen receptor". Hormone and Metabolic Research. 24 (8): 367–70. doi:10.1055/s-2007-1003337. PMID 1526623. S2CID 33271618. Archived from the original on 11 February 2020. Retrieved 4 July 2019.
- ^ Loose DS, Stover EP, Feldman D (July 1983). "Ketoconazole binds to glucocorticoid receptors and exhibits glucocorticoid antagonist activity in cultured cells". The Journal of Clinical Investigation. 72 (1): 404–8. doi:10.1172/jci110982. PMC 1129197. PMID 6135709.
- ^ Duret C, Daujat-Chavanieu M, Pascussi JM, Pichard-Garcia L, Balaguer P, Fabre JM, et al. (July 2006). "Ketoconazole and miconazole are antagonists of the human glucocorticoid receptor: consequences on the expression and function of the constitutive androstane receptor and the pregnane X receptor". Molecular Pharmacology. 70 (1): 329–39. doi:10.1124/mol.105.022046. PMID 16608920. S2CID 21455699.
- ^ Shaw JC (November 1996). "Antiandrogen therapy in dermatology". International Journal of Dermatology. 35 (11): 770–8. doi:10.1111/j.1365-4362.1996.tb02970.x. PMID 8915726. S2CID 39334280.
- ^ a b c d e f Sonino N (August 1986). "The endocrine effects of ketoconazole". Journal of Endocrinological Investigation. 9 (4): 341–7. doi:10.1007/BF03346939. PMID 3537102. S2CID 9148909.
- ^ a b Wheeler CJ, Keye WR, Peterson CM (2010). "Polycystic Ovary Syndrome". Reproductive Endocrinology and Infertility. pp. 147–182. doi:10.1007/978-1-4419-1436-1_11. ISBN 978-1-4419-1435-4.
- ^ a b c Drobnis EZ, Nangia AK (2017). "Antimicrobials and Male Reproduction". Impacts of Medications on Male Fertility. Advances in Experimental Medicine and Biology. Vol. 1034. pp. 131–161. doi:10.1007/978-3-319-69535-8_10. ISBN 978-3-319-69534-1. PMID 29256130.
- ^ Feldman D (November 1986). "Ketoconazole and other imidazole derivatives as inhibitors of steroidogenesis". Endocrine Reviews. 7 (4): 409–20. doi:10.1210/edrv-7-4-409. PMID 3536461.
- ^ Gal M, Orly J, Barr I, Algur N, Boldes R, Diamant YZ (May 1994). "Low dose ketoconazole attenuates serum androgen levels in patients with polycystic ovary syndrome and inhibits ovarian steroidogenesis in vitro". Fertility and Sterility. 61 (5): 823–32. doi:10.1016/S0015-0282(16)56691-6. PMID 8174717.
- ^ a b Lønning PE (2009). "New endocrine drugs for treatment of advanced breast cancer". Acta Oncologica. 29 (3): 379–86. doi:10.3109/02841869009090018. PMID 2194539.
- ^ Tarbit MH, Robertson WR, Lambert A (1990). "Hepatic and Endocrine Effects of Azole Antifungal Agents". Chemotherapy of Fungal Diseases. Handbook of Experimental Pharmacology. Vol. 96. pp. 205–229. doi:10.1007/978-3-642-75458-6_10. ISBN 978-3-642-75460-9. ISSN 0171-2004.
- ^ Philippaert K, Kerselaers S, Voets T, Vennekens R (April 2018). "2+-Activated Monovalent Cation-Selective Channels". SLAS Discovery. 23 (4): 341–352. doi:10.1177/2472555217748932. PMID 29316407.
- ^ Chin TW, Loeb M, Fong IW (August 1995). "Effects of an acidic beverage (Coca-Cola) on absorption of ketoconazole". Antimicrobial Agents and Chemotherapy. 39 (8): 1671–5. doi:10.1128/AAC.39.8.1671. PMC 162805. PMID 7486898.
- ^ a b c d e Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 720–. ISBN 978-1-4757-2085-3. Archived from the original on 10 January 2023. Retrieved 11 April 2017.
- ^ a b c d e f Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 586–. ISBN 978-3-88763-075-1. Archived from the original on 10 January 2023. Retrieved 22 July 2018.
- ^ Heeres J, Backx LJ, Mostmans JH, Van Cutsem J (August 1979). "Antimycotic imidazoles. part 4. Synthesis and antifungal activity of ketoconazole, a new potent orally active broad-spectrum antifungal agent". Journal of Medicinal Chemistry. 22 (8): 1003–5. doi:10.1021/jm00194a023. PMID 490531.
- ^ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Elsevier. pp. 1997–. ISBN 978-0-8155-1856-3. Archived from the original on 10 January 2023. Retrieved 14 June 2019.
- ^ a b Golan DE (2008). Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. Lippincott Williams & Wilkins. pp. 624–. ISBN 978-0-7817-8355-2. Archived from the original on 10 January 2023. Retrieved 11 April 2017.
- ^ "Ketoconazole-containing medicines". European Medicines Agency (EMA). 25 July 2013. Archived from the original on 22 February 2024. Retrieved 22 February 2024.
- ^ "Ketoconazole HRA recommended for approval in Cushing's syndrome". European Medicines Agency (EMA). 26 September 2014. Archived from the original on 22 February 2024. Retrieved 22 February 2024.
- ^ a b Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 159–. ISBN 978-94-011-4439-1.
- ^ a b c "Ketoconazole". Archived from the original on 23 July 2018. Retrieved 22 July 2018.
- ^ "European Medicines Agency recommends suspension of marketing authorisations for oral ketoconazole" (Press release). European Medicines Agency (EMA). 26 July 2013. Archived from the original on 14 January 2014.
- ^ "Levoketoconazole - Strongbridge Biopharma - AdisInsight". Archived from the original on 23 January 2020. Retrieved 14 June 2019.
- ^ Fleseriu M, Castinetti F (December 2016). "Updates on the role of adrenal steroidogenesis inhibitors in Cushing's syndrome: a focus on novel therapies". Pituitary. 19 (6): 643–653. doi:10.1007/s11102-016-0742-1. PMC 5080363. PMID 27600150.
- ^ KuKanich B (January 2008). "A review of selected systemic antifungal drugs for use in dogs and cats". Veterinary Medicine. Archived from the original on 5 October 2013.
| Antibiotics | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Arsenic compounds | |||||||||
| Quinoline derivatives | |||||||||
| Organic acids | |||||||||
| Sulfonamides | |||||||||
| Antifungals |
| ||||||||
| Other | |||||||||
Other dermatological preparations (D11) | |
|---|---|
| Anti-seborrheics | |
| Skin lightening | |
| Skin darkening | |
| Anti-inflammatories | |
| Alopecia treatments | |
| Hair growth inhibitors | |
| Others |
|
| Androgens (incl. AAS) |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antiandrogens |
| ||||||||||||
| |||||||||||||
| Glucocorticoids |
| ||||
|---|---|---|---|---|---|
| Antiglucocorticoids |
| ||||
| Synthesis modifiers | |||||
| |||||