 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C16H25NO |
| Molar mass | 247.382 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
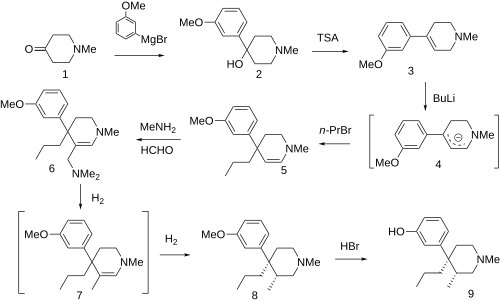
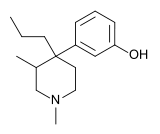
Picenadol (LY-97435) is a 4-phenylpiperidine derivative that is an opioid analgesic drug developed by Eli Lilly in the 1970s.[1]
Picenadol is an effective analgesic with similar efficacy to pethidine (meperidine). It has been investigated for some applications such as obstetrics[2] and dentistry,[3] but never commercialised.
It is unusual in that one enantiomer is a pure μ-opioid agonist, while the other is an antagonist.[4] The (3S,4R) isomer is the agonist, while (3R,4S) is antagonist.[5] This means that the racemic mix of the two enantiomers is a mixed agonist-antagonist, with relatively low abuse potential, and little of the κ-opioid activity that tends to cause problems with other opioid mixed agonist-antagonists such as pentazocine.[6]