 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
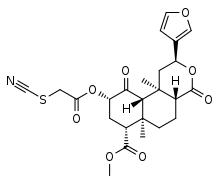
| Formula | C24H27NO8S |
| Molar mass | 489.54 g·mol−1 |
| 3D model (JSmol) | |
| |
RB-64 is a semi-synthetic derivative of salvinorin A. It is an irreversible agonist, with a reactive thiocyanate group that forms a bond to the κ-opioid receptor (KOR), resulting in very high potency.[1] It is functionally selective, activating G proteins more potently than β-arrestin-2.[2] RB-64 has a bias factor of up to 96 and is analgesic with fewer of the side-effects associated with unbiased KOR agonists.[3] The analgesia is long-lasting. Compared with unbiased agonists, RB-64 evokes considerably less receptor internalization.
See also
[edit]References
[edit]- ^ Yan F, Bikbulatov RV, Mocanu V, Dicheva N, Parker CE, Wetsel WC, et al. (July 2009). "Structure-based design, synthesis, and biochemical and pharmacological characterization of novel salvinorin A analogues as active state probes of the kappa-opioid receptor". Biochemistry. 48 (29): 6898–6908. doi:10.1021/bi900605n. PMC 2752672. PMID 19555087.
- ^ White KL, Scopton AP, Rives ML, Bikbulatov RV, Polepally PR, Brown PJ, et al. (January 2014). "Identification of novel functionally selective κ-opioid receptor scaffolds". Molecular Pharmacology. 85 (1): 83–90. doi:10.1124/mol.113.089649. PMC 3868907. PMID 24113749.
- ^ White KL, Robinson JE, Zhu H, DiBerto JF, Polepally PR, Zjawiony JK, et al. (January 2015). "The G protein-biased κ-opioid receptor agonist RB-64 is analgesic with a unique spectrum of activities in vivo". The Journal of Pharmacology and Experimental Therapeutics. 352 (1): 98–109. doi:10.1124/jpet.114.216820. PMC 4279099. PMID 25320048.