 | |
| Identifiers | |
|---|---|
| |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
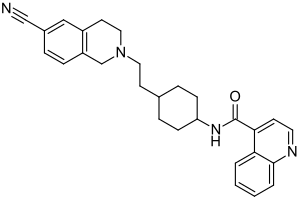
| Formula | C28H30N4O |
| Molar mass | 438.575 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
SB-277,011A is a drug which acts as a potent and selective dopamine D3 receptor antagonist,[1] which is around 80-100x selective for D3 over D2,[2] and lacks any partial agonist activity.[3]
SB-277,011A is used in the study of addiction to stimulant drugs such as nicotine[4] and cocaine.[5][6] Where cocaine reduces the threshold for brain electrical self-stimulation in rats, an indication of cocaine's rewarding effects, SB-277,011A completely reverses this effect. It may thus be useful in the treatment of addiction to nicotine and cocaine,[7][8][9] and is also being investigated for potential uses in the treatment of other drug addictions, such as addiction to heroin[10] and alcohol.[11][12][13]
Another potential application for SB-277,011A is in the treatment of schizophrenia,[14] and it may be particularly useful in treating comorbid patients who are both schizophrenic and addicted to drugs.[15] However it may worsen side effects such as tardive dyskinesia in patients who are already prescribed antipsychotic drugs.[16]