| |
| Names | |
|---|---|
| IUPAC name
9,20,21,25-tetramethoxy-15,30-dimethyl-7,23-dioxa-15,30-diazaheptacyclo[22.6.2.23,6.18,12.114,18.027,31.022,33]hexatriaconta-3,5,8(34),9,11,18,20,22(33),24(32),25,27(31),35-dodecaene
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ECHA InfoCard | 100.208.615 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C38H42N2O6 | |
| Molar mass | 622.74988 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
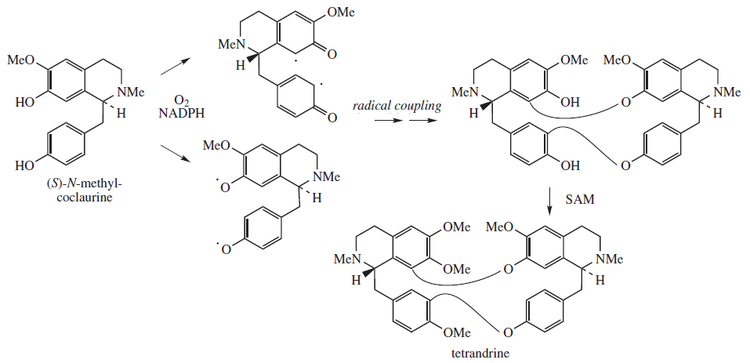
Tetrandrine, a bis-benzylisoquinoline alkaloid, is a calcium channel blocker.[1] It is isolated from the plant Stephania tetrandra,[2] and other Chinese and Japanese herbs.