 | |
| Clinical data | |
|---|---|
| Trade names | Tonocard |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601248 |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | 0.9-1 (oral) |
| Protein binding | 10-20% |
| Metabolism | glucuronidation (primary) |
| Elimination half-life | 9-14 R, 13-20 S |
| Excretion | 30-50% urine (unchanged) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.050.441 |
| Chemical and physical data | |
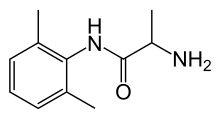
| Formula | C11H16N2O |
| Molar mass | 192.262 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Tocainide (Tonocard) is a class Ib antiarrhythmic agent. It is no longer sold in the United States.
