| Type | Whisky |
|---|---|
| Alcohol by volume | At least 40% |
| Proof (US) | At least 80° |
| Colour | Clear to off-white depending on ingredients |
| Ingredients | Grain (mashing), sugar (fermented water, kilju) |

Moonshine is high-proof liquor, traditionally made or distributed illegally.[1][2][3] Its clandestine distribution is known as bootlegging.[4] The name was derived from a tradition of distilling the alcohol at night to avoid detection. In the first decades of the 21st century, commercial distilleries have adopted the term for its outlaw cachet and begun producing their own legal "moonshine", including many novelty flavored varieties, that are said to continue the tradition by using a similar method and/or locale of production.[5]
Terminology
[edit]Different languages and countries have their own terms for moonshine (see Moonshine by country).
In English, moonshine is also known as mountain dew, choop, hooch (abbreviation of hoochinoo, name of a specific liquor, from Tlingit), homebrew, mulekick, shine, sneaky pete, white dog, white lightning, white/corn liquor, white/corn whiskey, pass around, firewater, and bootleg.[6][5]
Fractional crystallization
[edit]The ethanol may be concentrated in fermented beverages by means of freezing. For example, the name applejack derives from the traditional method of producing the drink, jacking, the process of freezing fermented cider and then removing the ice, increasing the alcohol content.[7][8] Starting with the fermented juice, with an alcohol content of less than ten percent, the concentrated result can contain 25–40% alcohol by volume (ABV).[9]
Moonshine stills
[edit]
In some countries, moonshine stills are illegal to sell, import, and own without permission. However, enthusiasts explain on internet forums how to obtain equipment and assemble it into a still.[10] To cut costs, stainless steel vessels are often replaced with plastic stills, vessels made from polypropylene that can withstand relatively high heat.
- A column still, or a spiral still, can achieve a vapor alcohol content of 95% ABV.
- Moonshine is usually distilled to 40% ABV, and seldom above 66% based on 48 samples.[11] For example, conventional pot stills commonly produce 40% ABV, and top out between 60% and 80% ABV after multiple distillations. However, ethanol can be dried to 95% ABV by heating 3A molecular sieves such as 3A zeolite.[12][13][14][15][16]
The preferred heat source for plastic stills or spiral stills is sous vide sticks; these control temperature, time, and circulation, and are therefore preferred over immersion heaters. Multiple units can be used to increase the wattage. Also, sous vide sticks, commonly sold in 1200 W and generally temperature regulated up to 90 °C (194 °F) (ethanol boils at 78 °C (172 °F)), will evaporate the ethanol faster than an immersion heater, commonly sold in 300 W. Electrical injury may occur if immersion heaters are modified, as if a 35 °C (95 °F) thermostat is removed from an aquarium heater (because doing so may break its waterproofing), or if an immersion heater is disassembled from an electric water boiler.
Evaporation stills
[edit]Plastic still
[edit]A plastic still is a device for distillation specially adapted for separating ethanol and water.[citation needed] Plastic stills are common because they are cheap and easy to manufacture. The principle is that a smaller amount of liquid is placed in an open smaller vessel inside a larger one that is closed. A cheap 100 W immersion heater is typically used as heat source, but a thermal immersion circulator, like a sous vide stick is ideal because it comes with a temperature controller. The liquid is kept heated at about 50 °C (122 °F) which slowly evaporates the ethanol to 40% ABV that condense on the inner walls of the outer vessel. The condensation that accumulates in the bottom of the vessel can then be diverted directly down through a filter containing activated carbon. The final product has approximately twice as much alcohol content as the starting liquid and can be distilled several times if stronger distillate is desired. The method is slow, and is not suitable for large-scale production.
Boiling stills
[edit]Fractional distillation
[edit]Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. It uses distillation to fractionate. Generally the component parts have boiling points that differ by less than 25 °C (45 °F) from each other under a pressure of one atmosphere.
Column still
[edit]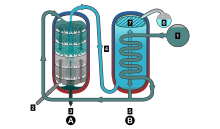
- Analyzer*
- Rectifier*
- Wash
- Steam
- Liquid out
- Alcohol vapor
- Recycled less volatile components
- Most volatile components
- Condenser
A column still, also called a continuous still, patent still or Coffey still, is a variety of still consisting of two columns. A column still can achieve a vapor alcohol content of 95% ABV.
Spiral still
[edit]A spiral still is a type of column still with a simple slow air-cooled distillation apparatus, commonly used for bootlegging.[10] Column and cooler consist of a 5-foot-long (1.5 m) copper tube wound in spiral form. The tube first goes up to act as a simple column, and then down to cool the product. Cookware usually consists of a 30-litre (6.6 imp gal; 7.9 US gal) plastic wine bucket. The heat source is typically a thermal immersion circulator (commonly runs at 1200 W), like a sous vide stick because it is hard to find 300 W immersion heaters, and it is risky to disassemble the immersion heater from an electric water boiler because it may cause electrical injury. The spiral burner is popular because, despite its simple construction and low manufacturing cost, it can provide 95% ABV.
Pot still
[edit]A pot still is a type of distillation apparatus or still used to distill flavored liquors such as whisky or cognac, but not rectified spirit because they are poor at separating congeners. Pot stills operate on a batch distillation basis (as opposed to a Coffey or column stills, which operate on a continuous basis). Traditionally constructed from copper, pot stills are made in a range of shapes and sizes depending on quantity and style of spirit. Geographic variations in still design exist, with certain kinds popular in parts of Appalachia, a region known for moonshine distilling.
Spirits distilled in pots commonly have 40% ABV, and top out between 60 and 80% after multiple distillations.
Safety
[edit]
Poorly produced moonshine can be contaminated, mainly from materials used in the construction of the still. Stills employing automotive radiators as condensers are particularly dangerous; in some cases, glycol produced from antifreeze can be a problem.
The head that comes immediately after the foreshot (the initial product of the still) typically contains small amounts of other undesirable compounds, such as acetone and various aldehydes.[17] Fusel alcohols are other undesirable byproducts of fermentation that are contained in the "aftershot," and are also typically discarded.
Alcohol concentrations at higher strengths (the GHS identifies concentrations above 24% ABV as dangerous[18]) are flammable and therefore dangerous to handle. This is especially true during the distilling process, when vaporized alcohol may accumulate in the air to dangerous concentrations if adequate ventilation is not provided.
Adulterated moonshine
[edit]The prolonged consumption of impure moonshine may cause renal disease, primarily from increased lead content.[19]
Lead
[edit]Radiators used as condensers may contain lead at the plumbing joints, and their use has resulted in blindness or lead poisoning[20] from tainted liquor.[21] This was a deadly hazard during the Prohibition-era United States. Consumption of lead-tainted moonshine is a serious risk factor for saturnine gout, a very painful but treatable medical condition that damages the kidneys and joints.[22][23]
Methanol
[edit]Contamination is still possible by unscrupulous distillers using cheap methanol to increase the apparent strength of the product. Moonshine can be made both more palatable and perhaps less dangerous by discarding the "foreshot" – the first 50–150 millilitres (1.8–5.3 imp fl oz; 1.7–5.1 US fl oz) of alcohol that drip from the condenser. Because methanol vaporizes at a lower temperature than ethanol, it is commonly believed that the foreshot contains most of the methanol, if any, from the mash. However, research shows that methanol is present until the very end of the distillation run.[24] Despite this, distillers will usually collect the foreshots until the temperature of the still reaches 80 °C (176 °F).[citation needed]
Outbreaks of methanol poisoning have occurred from methanol accidentally produced in moonshine production or deliberately used to strengthen it.[25]
Purification
[edit]In modern times, reducing methanol with the absorption of a molecular sieve is a practical method for production.[26]
Methanol safety by fermentation ingredient
[edit]- Grain starches: Methanol is not produced in toxic amounts by fermentation of sugars from grain starches.[citation needed]
- Methanol is produced in the fermentation of fruits via the action of pectin methylesterase, while of regulatory concern this does not typically exceed toxic levels and poisonings typically occur from methanol addition [27][28] (e.g. as a lacing agent).
Tests
[edit]Alcohols
[edit]
The Lucas test in alcohols is a test to differentiate between primary, secondary, and tertiary alcohols. It can be used to detect the levels of fusel alcohols.
Strength
[edit]A quick estimate of the alcoholic strength, or proof, of the distillate (the ratio of alcohol to water) is often achieved by shaking a clear container of the distillate. Large bubbles with a short duration indicate a higher alcohol content, while smaller bubbles that disappear more slowly indicate lower alcohol content.[citation needed]
A more reliable method is to use an alcoholmeter or hydrometer. A hydrometer is used during and after the fermentation process to determine the potential alcohol percentage of the moonshine, whereas an alcoholmeter is used after the product has been distilled to determine the volume percent or proof.[citation needed]
Misconceptions
[edit]
A common folk test for the quality of moonshine was to pour a small quantity of it into a spoon and set it on fire. The theory was that a safe distillate burns with a blue flame, but a tainted distillate burns with a yellow flame. Practitioners of this simple test also held that if a radiator coil had been used as a condenser, then there would be lead in the distillate, which would give a reddish flame. This led to the mnemonic, "Lead burns red and makes you dead," or simply, "Red means dead."[29][unreliable medical source?]
Legality
[edit]Manufacturing of spirits through distilling, fractional crystallization, etc. outside a registered distillery is illegal in many countries. Currently in the United States, there are four states that allow the production of moonshine for personal consumption (Alaska, Arizona, Massachusetts, and Missouri). Additionally, North Dakota law permits the production of moonshine for personal consumption up to the federally legal amount—which is zero gallons; entailing that production of any amount is illegal.[30]
| States | Legality |
|---|---|
| Alaska | Personal use only |
| Arizona | Must have a permit for personal use/register still |
| Massachusetts | Personal use on own property only |
| Missouri | Personal use up to 200 gal/year |
| North Dakota | Only up to federally-allowed amount (zero gallon) |
Society and culture
[edit]Popular offerings for the Maya deity and folk saint Maximón include money, tobacco, and moonshine.[31]
History
[edit]


Moonshine is by tradition usually a clear, unaged whiskey,[32] once made with barley mash in Scotland and Ireland or maize corn mash in the United States,[33] though plain sugar became just as common in illicit liquor during the last century. The word originated in the British Isles as a result of excise laws and became meaningful in the United States only after a tax passed after the Revolutionary War outlawing non-registered stills with the Tariff of 1791, or the Excise Whiskey Tax of 1791. This tax led to the Whiskey Rebellion lasting between 1791 and 1794, where the tax went unpaid by the rebels and was met with violent protest. This tax lasted until 1802, when it was repealed.[34] Another tax was introduced with the Revenue Act of 1861 and Revenue Act of 1862 that heavily taxed the production of spirits leading to an increase in illegal distilling and in turn, increased action from revenue agents that enforced the taxes.[6] Illegal distilling accelerated during the Prohibition era (1920–1933), which mandated a total ban on alcohol production under the Eighteenth Amendment of the Constitution. Since the amendment was repealed in 1933, laws focus on evasion of taxation on any type of spirits or intoxicating liquors. Applicable laws were historically enforced by the Bureau of Alcohol, Tobacco, Firearms and Explosives of the US Department of Justice, but are now usually handled by state agencies. Enforcement agents were once known colloquially as "revenuers".
Etymology
[edit]The earliest known instance of the term "moonshine" being used to refer to illicit alcohol dates to the 1785 edition of Grose's Dictionary of the Vulgar Tongue, which was published in England. Prior to that, "moonshine" referred to anything "illusory" or to literally the light of the moon.[35] The U.S. Government considers the word a "fanciful term" and does not regulate its use on the labels of commercial products, as such, legal moonshines may be any type of spirit, which must be indicated elsewhere on the label.[36]
Prohibition in the United States
[edit]In Prohibition-era United States, moonshine distillation was done at night to deter discovery.[37] While moonshiners were present in urban and rural areas around the United States after the Civil War, moonshine production concentrated in Appalachia because the limited road network made it easy to evade revenue officers and because it was difficult and expensive to transport corn crops. As a study of farmers in Cocke County, Tennessee, observes: "One could transport much more value in corn if it was first converted to whiskey. One horse could haul ten times more value on its back in whiskey than in corn."[38] Moonshiners such as Maggie Bailey of Harlan County, Kentucky, Amos Owens of Rutherford County, North Carolina, and Marvin "Popcorn" Sutton of Maggie Valley, North Carolina, became legendary.[39][40]
Once the liquor was distilled, drivers called "runners" or "bootleggers" smuggled moonshine liquor across the region in cars specially modified for speed and load-carrying capacity.[41] The cars were ordinary on the outside but modified with souped-up engines, extra interior room, and heavy-duty shock absorbers to support the weight of the illicit alcohol. After Prohibition ended, the out-of-work drivers kept their skills sharp through organized races, which led to the formation of the National Association for Stock Car Auto Racing (NASCAR).[42] Several former "runners," such as Junior Johnson, became noted drivers in the sport.[41]
Some varieties of maize corn grown in the United States were once prized for their use in moonshine production. One such variety used in moonshine, Jimmy Red corn, a "blood-red, flint-hard 'dent' corn with a rich and oily germ," almost became extinct when the last grower died in 2000. Two ears of Jimmy Red were passed on to "seed saver" Ted Chewning, who saved the variety from extinction and began to produce it on a wider scale.[43]
There have been modern-day attempts on the state level to legalize home distillation of alcohol, similar to how some states have been treating cannabis, despite there being federal laws prohibiting the practice. For example, the New Hampshire state legislature has tried repeatedly to pass laws allowing unlicensed home distillation of small batches.[44] In 2023, Ohio introduced legislation to do the same, with other states likely to follow.[45]
See also
[edit]- Bootleggers and Baptists
- Bureau of Alcohol, Tobacco, Firearms and Explosives (ATF)
- Congener (alcohol)
- Dixie Mafia
- Farmhouse ale
- Free Beer
- Homebrewing
- Moonshine by country
- Moonshine in popular culture
- Nip joint
- Rum-running
- Sour mash
References
[edit]- ^ Kosar, Kevin (15 April 2017). Moonshine: A Global History. London. ISBN 978-1-78023-742-8.
((cite book)): CS1 maint: location missing publisher (link) - ^ "moonshine". dictionary.com. Retrieved 26 March 2023.
- ^ "moonshine". Cambridge Dictionary. Retrieved 26 March 2023.
- ^ "bootleg". dictionary.com. Retrieved 26 March 2023.
- ^ a b Modern Moonshine : The Revival of White Whiskey in the Twenty-First Century. Cameron D. Lippard, Bruce E. Stewart (First ed.). Morgantown: West Virginia University Press. 2019. ISBN 978-1-946684-83-7. OCLC 1050142447.
((cite book)): CS1 maint: others (link) - ^ a b Joyce, Jaime (10 June 2014). Moonshine : a cultural history of America's infamous liquor. MBI Publishing Company. ISBN 978-0-7603-4584-9. OCLC 1242988394.
- ^ Ken Albala (2010). "Applejack". In Rachel Black (ed.). Alcohol in Popular Culture: An Encyclopedia. Santa Barbara, CA: ABC-CLIO. p. 10. ISBN 978-0-313-38048-8.
- ^ Michael Foley, Drinking with the Saints: The Sinner's Guide to a Holy Happy Hour (2015, ISBN 1621573834): Perhaps the most interesting option is applejack, the first distilled liquor native to North America and a great favorite among the colonists. [Now] usually a blend of apple brandy and neutral spirits that retains the flavor of the apples[.]
- ^ Sanborn Conner Brown, Wines & Beers of Old New England: A How-to-do-it History (1978, ISBN 0874511488)
- ^ a b "Spiralbrännaren" (PDF) (in Swedish).
- ^ Holstege, CP; Ferguson, JD; Wolf, CE; Baer, AB; Poklis, A (2004). "Analysis of Moonshine for Contaminants". Journal of Toxicology. Clinical Toxicology. 42 (5): 597–601. doi:10.1081/clt-200026976. PMID 15462151. S2CID 97866750.
- ^ Carmo, M. J.; Gubulin, J. C. (September 1997). "Ethanol-Water Adsorption on Commercial 3A Zeolites: Kinetic and Thermodynamic Data". Brazilian Journal of Chemical Engineering. 14 (3). doi:10.1590/S0104-66321997000300004. ISSN 0104-6632.
- ^ Burfield, David R.; Hefter, Glenn T.; Koh, Donald S. P. (1984). "Desiccant efficiency in solvent and reagent drying 8. molecular sieve column drying of 95% ethanol: An application of hygrometry to the assay of solvent water content". Journal of Chemical Technology and Biotechnology. Chemical Technology. 34 (4): 187–194. Bibcode:1984JCTBC..34..187B. doi:10.1002/jctb.5040340408.
- ^ Simo, Marian; Sivashanmugam, Siddharth; Brown, Christopher J.; Hlavacek, Vladimir (21 October 2009). "Adsorption/Desorption of Water and Ethanol on 3A Zeolite in Near-Adiabatic Fixed Bed". Industrial & Engineering Chemistry Research. 48 (20): 9247–9260. doi:10.1021/ie900446v.
- ^ Simo, Marian; Sivashanmugam, Siddharth; Brown, Christopher J.; Hlavacek, Vladimir (21 October 2009). "Adsorption/Desorption of Water and Ethanol on 3A Zeolite in Near-Adiabatic Fixed Bed". Industrial & Engineering Chemistry Research. 48 (20): 9247–9260. doi:10.1021/ie900446v.
- ^ Burfield, David R.; Hefter, Glenn T.; Koh, Donald S. P. (1984). "Desiccant efficiency in solvent and reagent drying 8. molecular sieve column drying of 95% ethanol: An application of hygrometry to the assay of solvent water content". Journal of Chemical Technology and Biotechnology. Chemical Technology. 34 (4): 187–194. Bibcode:1984JCTBC..34..187B. doi:10.1002/jctb.5040340408.
- ^ "Making Moonshine: The Dummies' Guide". Copper Moonshine Still Kits - Clawhammer Supply. Retrieved 25 November 2018.
- ^ "Hazardous Goods Management". Retrieved 31 August 2017.
- ^ "Risk of End Stage Renal Disease Associated with Alcohol Consumption" (PDF). Oxford Journals. Archived from the original (PDF) on 20 October 2016. Retrieved 24 December 2011.
- ^ "Why Your Copper Moonshine Still Needs To Be Lead Free – Whiskey Still Company". December 2016.
- ^ Peine & Schafft 2012, p. 97.
- ^ Dalvi, Sam R.; Pillinger, Michael H. (May 2013). "Saturnine gout, redux: a review". The American Journal of Medicine. 126 (5): 450.e1–8. doi:10.1016/j.amjmed.2012.09.015. ISSN 1555-7162. PMID 23510947.
- ^ Loghman-Adham M (September 1997). "Renal effects of environmental and occupational lead exposure". Environ. Health Perspect. 105 (9): 928–938. doi:10.2307/3433873. JSTOR 3433873. PMC 1470371. PMID 9300927.
- ^ Spaho, Nermina (28 June 2017). "Distillation Techniques in the Fruit Spirits Production". Distillation - Innovative Applications and Modeling. doi:10.5772/66774. ISBN 978-953-51-3201-1.
- ^ "Application to Include Fomepizole on the WHO Model List of Essential Medicines" (PDF). November 2012. p. 10.
- ^ Hui-Ling Ma; Xiu-Ping Yang; Ying Zuo (15 April 2006). "Study on Method of Decreasing Methanol in Apple Pomace Spirit". Food Science. 27 (4): 138–142.
- ^ Blumenthal, P; Steger, MC; Einfalt, D; Rieke-Zapp, J; Quintanilla Bellucci, A; Sommerfeld, K; Schwarz, S; Lachenmeier, DW (28 April 2021). "Methanol Mitigation during Manufacturing of Fruit Spirits with Special Consideration of Novel Coffee Cherry Spirits". Molecules. 26 (9): 2585. doi:10.3390/molecules26092585. PMC 8125215. PMID 33925245.
- ^ Lachenmeier, Dirk W.; Neufeld, Maria; Rehm, Jürgen (2021). "The impact of unrecorded alcohol use on health: What do we know in 2020?". J. Stud. Alcohol Drugs. 82: 28–41. doi:10.15288/jsad.2021.82.28. PMID 33573720.
- ^ "Moonshine". Skylark Medical Clinic. Archived from the original on 16 July 2011. Retrieved 23 July 2008.
- ^ "Moonshine Legal States 2023". worldpopulationreview.com. Retrieved 30 March 2023.
- ^ Meet Maximón: The Liquor-Drinking, Chain-Smoking Saint, National Geographic, Bethany Jones, January 24th, 2018
- ^ "Exploding moonshine: The new golden age of outlaw liquor". CNN. 17 June 2015. Retrieved 2 July 2017.
- ^ Guy Logsdon, Oklahoma Historical Society. "Moonshine". Encyclopedia of Oklahoma History & Culture. Oklahoma State University. Archived from the original on 9 October 2008. Retrieved 21 March 2014. Alt URL
- ^ "TTBGov - Whiskey Rebellion". www.ttb.gov. Retrieved 31 March 2023.
- ^ Kosar, Kevin (15 April 2017). Moonshine: A Global History. London. ISBN 978-1-78023-742-8. OCLC 1028980463.
((cite book)): CS1 maint: location missing publisher (link) - ^ Spoelman, Colin (22 October 2013). The Kings County Distillery Guide to Urban Moonshining : How to Make and Drink Whiskey. Haskell, David, 1979-. New York. ISBN 978-1-4197-0990-6. OCLC 843332480.
((cite book)): CS1 maint: location missing publisher (link) - ^ Sumich, Jason. "It's All Legal Until You Get Caught: Moonshining in the Southern Appalachians". Appalachian State University. Retrieved 21 March 2014.
- ^ Peine & Schafft 2012, pp. 98–99.
- ^ Block, Melissa (8 December 2005). "'Queen of the Mountain Bootleggers' Maggie Bailey". National Public Radio. Retrieved 4 May 2015.
- ^ Motsinger, Carol (10 November 2014). "New Movie Focuses on WNC Moonshiner Popcorn Sutton". Asheville Citizen-Times. Retrieved 16 May 2016.
- ^ a b Cooper, William J.; Terrill, Thomas E. (2009). The American South: A History, Volume II (4th ed.). Lanham, Md.: Rowman & Littlefield. p. 625. ISBN 978-0-7425-6097-0.
- ^ Billock, Jennifer. "How Moonshine Bootlegging Gave Rise to NASCAR". Smithsonian. Retrieved 4 April 2019.
- ^ Neimark, Jill (2 January 2018). "From Hooch To Haute Cuisine: A Nearly Extinct Bootlegger's Corn Gets A Second Shot". npr.org. NPR. Archived from the original on 23 June 2022. Retrieved 23 June 2022.
((cite web)): CS1 maint: bot: original URL status unknown (link) - ^ "Should Home Distilling Be Legal In N.H.? Lawmakers To Vote Wednesday". 2 January 2018.
- ^ "S.B. No. 13". Retrieved 30 January 2023.
Sources
[edit]- Davis, Elaine. Minnesota 13: "Wet" Wild Prohibition Days (2007) ISBN 978-0-9798017-0-9
- Peine, Emelie K.; Schafft, Kai A. (Spring–Fall 2012). "Moonshine, Mountaineers, and Modernity: Distilling Cultural History in the Southern Appalachian Mountains". Journal of Appalachian Studies. 18 (1/2). Appalachian Studies Association: 93–112. doi:10.2307/23337709. JSTOR 23337709. S2CID 142229259.
- Rowley, Matthew. Moonshine! History, songs, stories, and how-tos (2007) ISBN 978-1-57990-648-1
- Watman, Max. Chasing the White Dog: An Amateur Outlaw's Adventures in Moonshine (2010) ISBN 978-1-4391-7024-3
- King, Jeff. The Home Distiller's Workbook: Your Guide to Making Moonshine, Whisky, Vodka, Rum and So Much More! (2012) ISBN 978-1-4699-8939-6
External links
[edit]- "Moonshine – Blue Ridge Style" Archived 29 July 2012 at the Wayback Machine An Exhibition Produced by the Blue Ridge Institute and the Museum of Ferrum College
- Déantús an Phoitín (Poteen Making), by Mac Dara Ó Curraidhín (a one-hour 1998 Irish documentary film on the origins of the craft).
- North Carolina Moonshine – Historical information, images, music, and film excerpts
- Moonshine news page – Alcohol and Drugs History Society
- Georgia Moonshine Archived 23 May 2006 at the Wayback Machine – History and folk traditions in Georgia, USA
- "Moonshine 'tempts new generation'" – BBC on distilling illegal liquor in the 21st century.
- Moonshine Franklin Co Virginia Moonshine Still from the past – Video
- moonshine stills at a-c-e.uk
| |||||||||||
| |||||||||||
| |||||||||||
| |||||||||||
| By country | |
|---|---|
| By topic |
|
| People |
|
| Political parties | |
| Related | |
1776 to 1912 | |||||||||||||||||||||||||||||||||||||||||||
| Native Nations |
| ||||||||||||||||||||||||||||||||||||||||||
| Notable people |
| ||||||||||||||||||||||||||||||||||||||||||
| Frontier culture |
| ||||||||||||||||||||||||||||||||||||||||||
| Transport and trails | |||||||||||||||||||||||||||||||||||||||||||
| Folklore | |||||||||||||||||||||||||||||||||||||||||||
| Gold rushes | |||||||||||||||||||||||||||||||||||||||||||
| Gunfights | |||||||||||||||||||||||||||||||||||||||||||
| Military conflicts | |||||||||||||||||||||||||||||||||||||||||||
| Range wars and feuds | |||||||||||||||||||||||||||||||||||||||||||
| Lists | |||||||||||||||||||||||||||||||||||||||||||
| Influence | |||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||