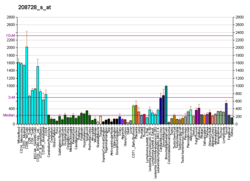
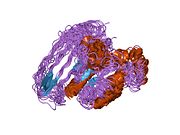
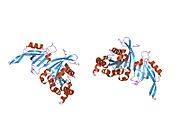
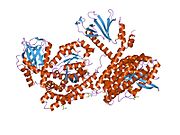
Cell division control protein 42 homolog (Cdc42 or CDC42) is a protein that in humans is encoded by the CDC42 gene. Cdc42 is involved in regulation of the cell cycle. It was originally identified in S. cerevisiae (yeast) as a mediator of cell division,[5][6] and is now known to influence a variety of signaling events and cellular processes in a variety of organisms from yeast to mammals.
Function
[edit]Human Cdc42 is a small GTPase of the Rho family, which regulates signaling pathways that control diverse cellular functions including cell morphology, cell migration, endocytosis and cell cycle progression.[7] Rho GTPases are central to dynamic actin cytoskeletal assembly and rearrangement that are the basis of cell-cell adhesion and migration. Activated Cdc42 activates by causing conformational changes[8] in p21-activated kinases PAK1 and PAK2, which in turn initiate actin reorganization and regulate cell adhesion, migration, and invasion.[9]
Structure
[edit]Cdc42 is a homodimer with A and B chains.[10] Its total length is 191 amino acids and its theoretical weight is 21.33 kDa.[10] Its sequence domains include a P-loop containing nucleoside triphosphate hydrolase and a small GTP-binding protein domain.[10]
Cdc42 cycles between an active GTP-bound state and an inactive GDP-bound state. This process is regulated by guanine nucleotide exchange factors (GEFs) which promote the exchange of bound GDP for free GTP, GTPase activating proteins (GAPs) which increase GTP hydrolysis activity, and GDP dissociation inhibitors which inhibit the dissociation of the nucleotide from the GTPase.[11]
Role in cancer
[edit]Recently, Cdc42 has been shown to actively assist in cancer progression. Several studies have established the basis for this and hypothesized about the underlying mechanisms.
Cdc42 is overexpressed in non-small cell lung cancer, colorectal adenocarcinoma, melanoma, breast cancer, and testicular cancer.[12] Elevated levels of the protein have been correlated with negative patient survival. Cdc42 has also been shown to be required for both G1-S phase progression and mitosis, and it also modulates the transcription factors SRF, STAT3, and NFkB.[12] It has been hypothesized that targeting Cdc42 in conjunction with chemotherapy may be an effective cancer treatment strategy.
In one study studying the role of Cdc42 in cervical cancer, immunohistochemistry was used to detect Cdc42 expression in three types of tissues: normal cervical tissues, cervical intraepithelial neoplasia (CIN) I or below, CIN II or above, and cervical cancer tissues.[13] Cdc42 expression was gradually increased showing significant difference and was significantly higher in HeLa cells than in regular cells. The migration ability of HeLa cells transfected with Cdc42 was higher than that of non-transfected cells.[13] It was proposed that the overexpression of Cdc42 can promote filopodia formation in HeLa cells. Cdc42 overexpression significantly improved the ability of cervical cancer cells to migrate, possibly due to improved pseudopodia formation.[13]
Another study found that Cdc42 drives the process of initiating a metastatic tumor in a new tissue by promoting the expression of β1 integrin, an adhesion receptor known to be involved in metastasis.[14] Levels of β1 integrin were reduced in Cdc42-deficient cells. β1 integrin is important for adhesion to the extracellular matrix, and could be important for the initial attachment to endothelial cells as well. Knocking down β1 integrin inhibited cancer cell migration, whereas overexpressing the integrin in Cdc42-deficient cells restored endothelial invasion.[14] Cdc42 promoted β1 integrin expression by activating a transcription factor called SRF. A continually active form of the transcription factor was also capable of restoring endothelial insertion to cancer cells lacking Cdc42.
Normal cancer cells and Cdc42-deficient cancer cells have also been compared in vivo. When both types of cells were injected into mouse tail veins, control cells spread out more on the vessel endothelium within minutes, suggesting that Cdc42 assists in cell migration.[14] After six weeks, the control cells had generated more metastases than the Cdc42-deficient cells. Invading cancer cells send out protrusions that reach down between neighboring endothelial cells to contact the underlying basement membrane. The cancer cells then spread out on this extracellular matrix so that the endothelial cells retract, and allow the invaders to insert themselves between them.[14] In the absence of Cdc42, cancer cells failed to spread out on the basement membrane, and Cdc42-deficient cells showed reduced adhesion to extracellular matrix-coated coverslips.[14] Cdc42 therefore promotes the attachment of cancer cells to both endothelial cells and the underlying basement membrane during transendothelial migration.
The small molecular inhibitor AZA197 has been used to inhibit Cdc42 in the treatment of KRAS mutant colorectal cancers.[15] There was evidence that Cdc42 inhibition by AZA197 treatment suppresses proliferative and pro-survival signaling pathways via PAK1-ERK signaling and reduces colon cancer cell migration and invasion.[15] In mice, systemic AZA197 treatment in vivo reduced primary tumor growth and prolonged survival.[15] Therapy targeting Rho GTPase Cdc42 signaling pathways may be effective for treatment of patients with advanced colon cancer overexpressing Cdc42, and particularly those with KRAS-mutant disease.
Takenouchi-Kosaki syndrome
[edit]Mutations in the CDC42 gene are responsible for the Takenouchi-Kosaki syndrome, an autosomal-dominant complex congenital developmental disorder associated with thrombocytopenia.[16] Alleic variants that are associated with the syndrome include mutations in the CDC42 gene that are expressed at the protein level as Ile21Thr, Tyr64Cys, Arg66Gly, Cys81Phe, Ser83Pro, or Glu171Lys.[17]
Interactions
[edit]CDC42 has been shown to interact with:
- ARHGAP1,[18][19][20][21]
- ARHGDIA,[22][23]
- BAIAP2,[24][25][26]
- BNIP2,[20][27][28]
- BNIPL,[29]
- CDC42EP2,[30][31]
- CDC42EP3,[30][32]
- ERRFI1,[33]
- GDI1,[34]
- IQGAP1,[21][35][36][37][38]
- IQGAP2,[39]
- ITSN1,[40][41]
- MAP3K10,[18]
- MAP3K11,[18][42]
- PAK1,[21][43]
- PAK2,[21][44][45]
- PAK4.[22][45][46]
- PAK7,[46][47]
- PARD6A,[48][49][50]
- PARD6B,[48][49][51]
- Phospholipase D1,[52]
- RICS[53][54][55]
- TRIP10,[56][57]
- WASL,[58][59]
- Wiskott–Aldrich syndrome protein,[57][60][61][62]
See also
[edit]References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000070831 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000006699 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Adams AE, Johnson DI, Longnecker RM, Sloat BF, Pringle JR (July 1990). "CDC42 and CDC43, two additional genes involved in budding and the establishment of cell polarity in the yeast Saccharomyces cerevisiae". The Journal of Cell Biology. 111 (1): 131–142. doi:10.1083/jcb.111.1.131. PMC 2116161. PMID 2195038.
- ^ Johnson DI, Pringle JR (July 1990). "Molecular characterization of CDC42, a Saccharomyces cerevisiae gene involved in the development of cell polarity". The Journal of Cell Biology. 111 (1): 143–152. doi:10.1083/jcb.111.1.143. PMC 2116164. PMID 2164028.
- ^ Qadir MI, Parveen A, Ali M (October 2015). "Cdc42: Role in Cancer Management". Chemical Biology & Drug Design. 86 (4): 432–9. doi:10.1111/cbdd.12556. PMID 25777055. S2CID 33006765.
- ^ Bishop AL, Hall A (June 2000). "Rho GTPases and their effector proteins". The Biochemical Journal. 348 (2): 241–255. doi:10.1042/bj3480241. PMC 1221060. PMID 10816416.
- ^ Guo Y, Kenney SR, Muller CY, Adams S, Rutledge T, Romero E, Murray-Krezan C, Prekeris R, Sklar LA, Hudson LG, Wandinger-Ness A (October 2015). "R-Ketorolac Targets Cdc42 and Rac1 and Alters Ovarian Cancer Cell Behaviors Critical for Invasion and Metastasis". Molecular Cancer Therapeutics. 14 (10): 2215–27. doi:10.1158/1535-7163.MCT-15-0419. PMC 4596774. PMID 26206334.
- ^ a b c "PDB 1a4r structure summary". Protein Data Bank in Europe. EMBL-EBI. Retrieved 2016-04-22.
- ^ "CDC42 (cell division cycle 42 (GTP binding protein, 25kDa))". atlasgeneticsoncology.org. Archived from the original on 2016-03-31. Retrieved 2016-04-22.
- ^ a b Stengel K, Zheng Y (September 2011). "Cdc42 in oncogenic transformation, invasion, and tumorigenesis". Cellular Signalling. 23 (9): 1415–23. doi:10.1016/j.cellsig.2011.04.001. PMC 3115433. PMID 21515363.
- ^ a b c Ye H, Zhang Y, Geng L, Li Z (February 2015). "Cdc42 expression in cervical cancer and its effects on cervical tumor invasion and migration". International Journal of Oncology. 46 (2): 757–63. doi:10.3892/ijo.2014.2748. PMID 25394485.
- ^ a b c d e Wilson JM, Menkhaus P, Gustin BW (May 1987). "Volume and outcome of coronary artery bypass graft surgery". JAMA. 257 (18): 2434–5. doi:10.1001/jama.1987.03390180052010. PMID 3494856.
- ^ a b c Zins K, Gunawardhana S, Lucas T, Abraham D, Aharinejad S (2013-01-01). "Targeting Cdc42 with the small molecule drug AZA197 suppresses primary colon cancer growth and prolongs survival in a preclinical mouse xenograft model by downregulation of PAK1 activity". Journal of Translational Medicine. 11: 295. doi:10.1186/1479-5876-11-295. PMC 4222769. PMID 24279335.
- ^ Santoro C, Gaudino G, Torella A, Piluso G, Perrotta S, Miraglia Del Giudice E, Nigro V, Grandone A (December 2021). "Intermittent macrothrombocytopenia in a novel patient with Takenouchi-Kosaki syndrome and review of literature". European Journal of Medical Genetics. 64 (12): 104358. doi:10.1016/j.ejmg.2021.104358. PMID 34624555. S2CID 238529349.
- ^ "CDC42 Allelic Variants". Online Mendelian Inheritance in Man (OMIM).
- ^ a b c Nagata K, Puls A, Futter C, Aspenstrom P, Schaefer E, Nakata T, Hirokawa N, Hall A (January 1998). "The MAP kinase kinase kinase MLK2 co-localizes with activated JNK along microtubules and associates with kinesin superfamily motor KIF3". The EMBO Journal. 17 (1): 149–58. doi:10.1093/emboj/17.1.149. PMC 1170366. PMID 9427749.
- ^ Li R, Zhang B, Zheng Y (December 1997). "Structural determinants required for the interaction between Rho GTPase and the GTPase-activating domain of p190". The Journal of Biological Chemistry. 272 (52): 32830–5. doi:10.1074/jbc.272.52.32830. PMID 9407060.
- ^ a b Low BC, Lim YP, Lim J, Wong ES, Guy GR (November 1999). "Tyrosine phosphorylation of the Bcl-2-associated protein BNIP-2 by fibroblast growth factor receptor-1 prevents its binding to Cdc42GAP and Cdc42". The Journal of Biological Chemistry. 274 (46): 33123–30. doi:10.1074/jbc.274.46.33123. PMID 10551883.
- ^ a b c d Zhang B, Chernoff J, Zheng Y (April 1998). "Interaction of Rac1 with GTPase-activating proteins and putative effectors. A comparison with Cdc42 and RhoA". The Journal of Biological Chemistry. 273 (15): 8776–82. doi:10.1074/jbc.273.15.8776. PMID 9535855.
- ^ a b Ewing RM, Chu P, Elisma F, Li H, Taylor P, Climie S, McBroom-Cerajewski L, Robinson MD, O'Connor L, Li M, Taylor R, Dharsee M, Ho Y, Heilbut A, Moore L, Zhang S, Ornatsky O, Bukhman YV, Ethier M, Sheng Y, Vasilescu J, Abu-Farha M, Lambert JP, Duewel HS, Stewart II, Kuehl B, Hogue K, Colwill K, Gladwish K, Muskat B, Kinach R, Adams SL, Moran MF, Morin GB, Topaloglou T, Figeys D (2007). "Large-scale mapping of human protein-protein interactions by mass spectrometry". Molecular Systems Biology. 3 (1): 89. doi:10.1038/msb4100134. PMC 1847948. PMID 17353931.
- ^ Gorvel JP, Chang TC, Boretto J, Azuma T, Chavrier P (January 1998). "Differential properties of D4/LyGDI versus RhoGDI: phosphorylation and rho GTPase selectivity". FEBS Letters. 422 (2): 269–73. doi:10.1016/S0014-5793(98)00020-9. PMID 9490022. S2CID 10817327.
- ^ Soltau M, Richter D, Kreienkamp HJ (December 2002). "The insulin receptor substrate IRSp53 links postsynaptic shank1 to the small G-protein cdc42". Molecular and Cellular Neurosciences. 21 (4): 575–83. doi:10.1006/mcne.2002.1201. PMID 12504591. S2CID 572407.
- ^ Krugmann S, Jordens I, Gevaert K, Driessens M, Vandekerckhove J, Hall A (October 2001). "Cdc42 induces filopodia by promoting the formation of an IRSp53:Mena complex". Current Biology. 11 (21): 1645–55. Bibcode:2001CBio...11.1645K. doi:10.1016/S0960-9822(01)00506-1. PMID 11696321. S2CID 11290377.
- ^ Miki H, Yamaguchi H, Suetsugu S, Takenawa T (December 2000). "IRSp53 is an essential intermediate between Rac and WAVE in the regulation of membrane ruffling". Nature. 408 (6813): 732–5. Bibcode:2000Natur.408..732M. doi:10.1038/35047107. PMID 11130076. S2CID 4426046.
- ^ Low BC, Seow KT, Guy GR (May 2000). "Evidence for a novel Cdc42GAP domain at the carboxyl terminus of BNIP-2". The Journal of Biological Chemistry. 275 (19): 14415–22. doi:10.1074/jbc.275.19.14415. PMID 10799524.
- ^ Low BC, Seow KT, Guy GR (December 2000). "The BNIP-2 and Cdc42GAP homology domain of BNIP-2 mediates its homophilic association and heterophilic interaction with Cdc42GAP". The Journal of Biological Chemistry. 275 (48): 37742–51. doi:10.1074/jbc.M004897200. PMID 10954711.
- ^ Qin W, Hu J, Guo M, Xu J, Li J, Yao G, Zhou X, Jiang H, Zhang P, Shen L, Wan D, Gu J (August 2003). "BNIPL-2, a novel homologue of BNIP-2, interacts with Bcl-2 and Cdc42GAP in apoptosis". Biochemical and Biophysical Research Communications. 308 (2): 379–85. doi:10.1016/S0006-291X(03)01387-1. PMID 12901880.
- ^ a b Joberty G, Perlungher RR, Macara IG (October 1999). "The Borgs, a new family of Cdc42 and TC10 GTPase-interacting proteins". Molecular and Cellular Biology. 19 (10): 6585–97. doi:10.1128/MCB.19.10.6585. PMC 84628. PMID 10490598.
- ^ Hirsch DS, Pirone DM, Burbelo PD (January 2001). "A new family of Cdc42 effector proteins, CEPs, function in fibroblast and epithelial cell shape changes". The Journal of Biological Chemistry. 276 (2): 875–83. doi:10.1074/jbc.M007039200. PMID 11035016.
- ^ Alberts AS, Bouquin N, Johnston LH, Treisman R (April 1998). "Analysis of RhoA-binding proteins reveals an interaction domain conserved in heterotrimeric G protein beta subunits and the yeast response regulator protein Skn7". The Journal of Biological Chemistry. 273 (15): 8616–22. doi:10.1074/jbc.273.15.8616. PMID 9535835.
- ^ Makkinje A, Quinn DA, Chen A, Cadilla CL, Force T, Bonventre JV, Kyriakis JM (June 2000). "Gene 33/Mig-6, a transcriptionally inducible adapter protein that binds GTP-Cdc42 and activates SAPK/JNK. A potential marker transcript for chronic pathologic conditions, such as diabetic nephropathy. Possible role in the response to persistent stress". The Journal of Biological Chemistry. 275 (23): 17838–47. doi:10.1074/jbc.M909735199. PMC 3668664. PMID 10749885.
- ^ Gibson RM, Wilson-Delfosse AL (October 2001). "RhoGDI-binding-defective mutant of Cdc42Hs targets to membranes and activates filopodia formation but does not cycle with the cytosol of mammalian cells". The Biochemical Journal. 359 (Pt 2): 285–94. doi:10.1042/0264-6021:3590285. PMC 1222146. PMID 11583574.
- ^ Kuroda S, Fukata M, Kobayashi K, Nakafuku M, Nomura N, Iwamatsu A, Kaibuchi K (September 1996). "Identification of IQGAP as a putative target for the small GTPases, Cdc42 and Rac1". The Journal of Biological Chemistry. 271 (38): 23363–7. doi:10.1074/jbc.271.38.23363. PMID 8798539.
- ^ Fukata M, Watanabe T, Noritake J, Nakagawa M, Yamaga M, Kuroda S, Matsuura Y, Iwamatsu A, Perez F, Kaibuchi K (June 2002). "Rac1 and Cdc42 capture microtubules through IQGAP1 and CLIP-170". Cell. 109 (7): 873–85. doi:10.1016/S0092-8674(02)00800-0. PMID 12110184. S2CID 15158637.
- ^ Hart MJ, Callow MG, Souza B, Polakis P (June 1996). "IQGAP1, a calmodulin-binding protein with a rasGAP-related domain, is a potential effector for cdc42Hs". The EMBO Journal. 15 (12): 2997–3005. doi:10.1002/j.1460-2075.1996.tb00663.x. PMC 450241. PMID 8670801.
- ^ Joyal JL, Annan RS, Ho YD, Huddleston ME, Carr SA, Hart MJ, Sacks DB (June 1997). "Calmodulin modulates the interaction between IQGAP1 and Cdc42. Identification of IQGAP1 by nanoelectrospray tandem mass spectrometry". The Journal of Biological Chemistry. 272 (24): 15419–25. doi:10.1074/jbc.272.24.15419. PMID 9182573.
- ^ Brill S, Li S, Lyman CW, Church DM, Wasmuth JJ, Weissbach L, Bernards A, Snijders AJ (September 1996). "The Ras GTPase-activating-protein-related human protein IQGAP2 harbors a potential actin binding domain and interacts with calmodulin and Rho family GTPases". Molecular and Cellular Biology. 16 (9): 4869–78. doi:10.1128/mcb.16.9.4869. PMC 231489. PMID 8756646.
- ^ Hussain NK, Jenna S, Glogauer M, Quinn CC, Wasiak S, Guipponi M, Antonarakis SE, Kay BK, Stossel TP, Lamarche-Vane N, McPherson PS (October 2001). "Endocytic protein intersectin-l regulates actin assembly via Cdc42 and N-WASP". Nature Cell Biology. 3 (10): 927–32. doi:10.1038/ncb1001-927. PMID 11584276. S2CID 22914248.
- ^ Snyder JT, Worthylake DK, Rossman KL, Betts L, Pruitt WM, Siderovski DP, Der CJ, Sondek J (June 2002). "Structural basis for the selective activation of Rho GTPases by Dbl exchange factors". Nature Structural Biology. 9 (6): 468–75. doi:10.1038/nsb796. PMID 12006984. S2CID 13161854.
- ^ Böck BC, Vacratsis PO, Qamirani E, Gallo KA (May 2000). "Cdc42-induced activation of the mixed-lineage kinase SPRK in vivo. Requirement of the Cdc42/Rac interactive binding motif and changes in phosphorylation". The Journal of Biological Chemistry. 275 (19): 14231–41. doi:10.1074/jbc.275.19.14231. PMID 10799501.
- ^ Seoh ML, Ng CH, Yong J, Lim L, Leung T (March 2003). "ArhGAP15, a novel human RacGAP protein with GTPase binding property". FEBS Letters. 539 (1–3): 131–7. doi:10.1016/S0014-5793(03)00213-8. PMID 12650940. S2CID 27574424.
- ^ Stevens WK, Vranken W, Goudreau N, Xiang H, Xu P, Ni F (May 1999). "Conformation of a Cdc42/Rac interactive binding peptide in complex with Cdc42 and analysis of the binding interface". Biochemistry. 38 (19): 5968–75. doi:10.1021/bi990426u. PMID 10320322.
- ^ a b Abo A, Qu J, Cammarano MS, Dan C, Fritsch A, Baud V, Belisle B, Minden A (November 1998). "PAK4, a novel effector for Cdc42Hs, is implicated in the reorganization of the actin cytoskeleton and in the formation of filopodia". The EMBO Journal. 17 (22): 6527–40. doi:10.1093/emboj/17.22.6527. PMC 1171000. PMID 9822598.
- ^ a b Dan C, Nath N, Liberto M, Minden A (January 2002). "PAK5, a new brain-specific kinase, promotes neurite outgrowth in N1E-115 cells". Molecular and Cellular Biology. 22 (2): 567–77. doi:10.1128/MCB.22.2.567-577.2002. PMC 139731. PMID 11756552.
- ^ Pandey A, Dan I, Kristiansen TZ, Watanabe NM, Voldby J, Kajikawa E, Khosravi-Far R, Blagoev B, Mann M (May 2002). "Cloning and characterization of PAK5, a novel member of mammalian p21-activated kinase-II subfamily that is predominantly expressed in brain". Oncogene. 21 (24): 3939–48. doi:10.1038/sj.onc.1205478. PMID 12032833. S2CID 281761.
- ^ a b Joberty G, Petersen C, Gao L, Macara IG (August 2000). "The cell-polarity protein Par6 links Par3 and atypical protein kinase C to Cdc42". Nature Cell Biology. 2 (8): 531–9. doi:10.1038/35019573. PMID 10934474. S2CID 27139234.
- ^ a b Noda Y, Takeya R, Ohno S, Naito S, Ito T, Sumimoto H (February 2001). "Human homologues of the Caenorhabditis elegans cell polarity protein PAR6 as an adaptor that links the small GTPases Rac and Cdc42 to atypical protein kinase C". Genes to Cells. 6 (2): 107–19. doi:10.1046/j.1365-2443.2001.00404.x. PMID 11260256.
- ^ Qiu RG, Abo A, Steven Martin G (June 2000). "A human homolog of the C. elegans polarity determinant Par-6 links Rac and Cdc42 to PKCzeta signaling and cell transformation". Current Biology. 10 (12): 697–707. Bibcode:2000CBio...10..697Q. doi:10.1016/S0960-9822(00)00535-2. PMID 10873802. S2CID 14825707.
- ^ Neudauer CL, Joberty G, Macara IG (January 2001). "PIST: a novel PDZ/coiled-coil domain binding partner for the rho-family GTPase TC10". Biochemical and Biophysical Research Communications. 280 (2): 541–7. doi:10.1006/bbrc.2000.4160. PMID 11162552.
- ^ Walker SJ, Wu WJ, Cerione RA, Brown HA (May 2000). "Activation of phospholipase D1 by Cdc42 requires the Rho insert region". The Journal of Biological Chemistry. 275 (21): 15665–8. doi:10.1074/jbc.M000076200. PMID 10747870.
- ^ Nakazawa T, Watabe AM, Tezuka T, Yoshida Y, Yokoyama K, Umemori H, Inoue A, Okabe S, Manabe T, Yamamoto T (July 2003). "p250GAP, a novel brain-enriched GTPase-activating protein for Rho family GTPases, is involved in the N-methyl-d-aspartate receptor signaling". Molecular Biology of the Cell. 14 (7): 2921–34. doi:10.1091/mbc.E02-09-0623. PMC 165687. PMID 12857875.
- ^ Zhao C, Ma H, Bossy-Wetzel E, Lipton SA, Zhang Z, Feng GS (September 2003). "GC-GAP, a Rho family GTPase-activating protein that interacts with signaling adapters Gab1 and Gab2". The Journal of Biological Chemistry. 278 (36): 34641–53. doi:10.1074/jbc.M304594200. PMID 12819203.
- ^ Nakamura T, Komiya M, Sone K, Hirose E, Gotoh N, Morii H, Ohta Y, Mori N (December 2002). "Grit, a GTPase-activating protein for the Rho family, regulates neurite extension through association with the TrkA receptor and N-Shc and CrkL/Crk adapter molecules". Molecular and Cellular Biology. 22 (24): 8721–34. doi:10.1128/MCB.22.24.8721-8734.2002. PMC 139861. PMID 12446789.
- ^ Aspenström P (July 1997). "A Cdc42 target protein with homology to the non-kinase domain of FER has a potential role in regulating the actin cytoskeleton". Current Biology. 7 (7): 479–87. Bibcode:1997CBio....7..479A. doi:10.1016/S0960-9822(06)00219-3. PMID 9210375. S2CID 14268690.
- ^ a b Tian L, Nelson DL, Stewart DM (March 2000). "Cdc42-interacting protein 4 mediates binding of the Wiskott–Aldrich syndrome protein to microtubules". The Journal of Biological Chemistry. 275 (11): 7854–61. doi:10.1074/jbc.275.11.7854. PMID 10713100.
- ^ Carlier MF, Nioche P, Broutin-L'Hermite I, Boujemaa R, Le Clainche C, Egile C, Garbay C, Ducruix A, Sansonetti P, Pantaloni D (July 2000). "GRB2 links signaling to actin assembly by enhancing interaction of neural Wiskott–Aldrich syndrome protein (N-WASp) with actin-related protein (ARP2/3) complex". The Journal of Biological Chemistry. 275 (29): 21946–52. doi:10.1074/jbc.M000687200. PMID 10781580.
- ^ Miki H, Sasaki T, Takai Y, Takenawa T (January 1998). "Induction of filopodium formation by a WASP-related actin-depolymerizing protein N-WASP". Nature. 391 (6662): 93–6. Bibcode:1998Natur.391...93M. doi:10.1038/34208. PMID 9422512. S2CID 4363890.
- ^ Kim AS, Kakalis LT, Abdul-Manan N, Liu GA, Rosen MK (March 2000). "Autoinhibition and activation mechanisms of the Wiskott–Aldrich syndrome protein". Nature. 404 (6774): 151–8. Bibcode:2000Natur.404..151K. doi:10.1038/35004513. PMID 10724160. S2CID 4416185.
- ^ Kolluri R, Tolias KF, Carpenter CL, Rosen FS, Kirchhausen T (May 1996). "Direct interaction of the Wiskott–Aldrich syndrome protein with the GTPase Cdc42". Proceedings of the National Academy of Sciences of the United States of America. 93 (11): 5615–8. Bibcode:1996PNAS...93.5615K. doi:10.1073/pnas.93.11.5615. PMC 39296. PMID 8643625.
- ^ Symons M, Derry JM, Karlak B, Jiang S, Lemahieu V, Mccormick F, Francke U, Abo A (March 1996). "Wiskott–Aldrich syndrome protein, a novel effector for the GTPase CDC42Hs, is implicated in actin polymerization". Cell. 84 (5): 723–34. doi:10.1016/S0092-8674(00)81050-8. PMID 8625410. S2CID 17838931.
External links
[edit]- cdc42+GTP-Binding+Protein at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- CDC42 Info with links in the Cell Migration Gateway
- Human CDC42 genome location and CDC42 gene details page in the UCSC Genome Browser.
| 3.6.1 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3.6.2 | |||||||||||||||
| 3.6.3-4: ATPase |
| ||||||||||||||
| 3.6.5: GTPase |
| ||||||||||||||
| Cyclin | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CDK | |||||||||
| CDK inhibitor | |||||||||
| P53 p63 p73 family | |||||||||
| Other | |||||||||
| Phases and checkpoints |
| ||||||||
| 3.6.1 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3.6.2 | |||||||||||||||
| 3.6.3-4: ATPase |
| ||||||||||||||
| 3.6.5: GTPase |
| ||||||||||||||