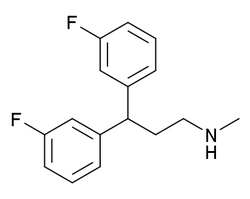
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C16H17F2N |
| Molar mass | 261.316 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Delucemine (NPS-1506) is a drug which acts as an NMDA antagonist and a serotonin reuptake inhibitor, and has neuroprotective effects.[1][2] It was originally investigated for the treatment of stroke and in 2004 was studied as a potential antidepressant.[3][4][5]