 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | IV, IM, SC, Other |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Liver |
| Excretion | Kidneys |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.034.559 |
| Chemical and physical data | |
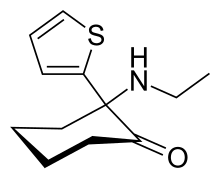
| Formula | C12H17NOS |
| Molar mass | 223.33 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Tiletamine is a dissociative anesthetic and pharmacologically classified as an NMDA receptor antagonist.[1] It is related chemically to ketamine.[2] Tiletamine hydrochloride exists as odorless white crystals.
It is used in veterinary medicine in the combination product Telazol (tiletamine/zolazepam, 50 mg/ml of each in 5 ml vial) as an injectable anesthetic for use in cats and dogs.[3][4][5] It is sometimes used in combination with xylazine (Rompun) to chemically immobilize large mammals such as polar bears[6] and wood bison.[7] Telazol is the only commercially available tiletamine product in the United States. It is contraindicated in patients of an ASA score of III or greater and in animals with CNS signs, hyperthyroidism, cardiac disease, pancreatic or renal disease, pregnancy, glaucoma, or penetrating eye injuries.[3]