 | |
| Clinical data | |
|---|---|
| Other names | MT-45, IC-6 |
| Routes of administration | oral administration rectal administration |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
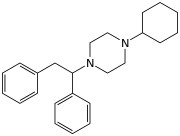
| Formula | C24H32N2 |
| Molar mass | 348.534 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
MT-45 (IC-6) is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co.[1] It is chemically a 1-substituted-4-(1,2-diphenylethyl) piperazine derivative, which is structurally unrelated to most other opioid drugs. Racemic MT-45 has around 80% the potency of morphine, with almost all opioid activity residing in the (S) enantiomer (the opposite stereochemistry from the related drug lefetamine).[2][3] It has been used as a lead compound from which a large family of potent opioid drugs[4] have been developed, including full agonists, partial agonists, and antagonists at the three main opioid receptor subtypes.[5][6][7][8][9][10] Fluorinated derivatives of MT-45 such as 2F-MT-45 are significantly more potent as μ-opioid receptor agonists, and one of its main metabolites 1,2-diphenylethylpiperazine also blocks NMDA receptors.[11]
