 | |
 | |
| Clinical data | |
|---|---|
| Dependence liability | Moderate |
| Routes of administration | Oral, sublingual, rectal |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
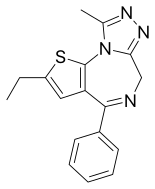
| Formula | C17H16N4S |
| Molar mass | 308.40 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Deschloroetizolam (also known as Etizolam-2) is a thienotriazolodiazepine that is the dechlorinated analog of the closely related etizolam.[1][2][3] The compound has been sold as a designer drug.[4][5][6][7]